The IUPAC name for the given hydrocarbon is:
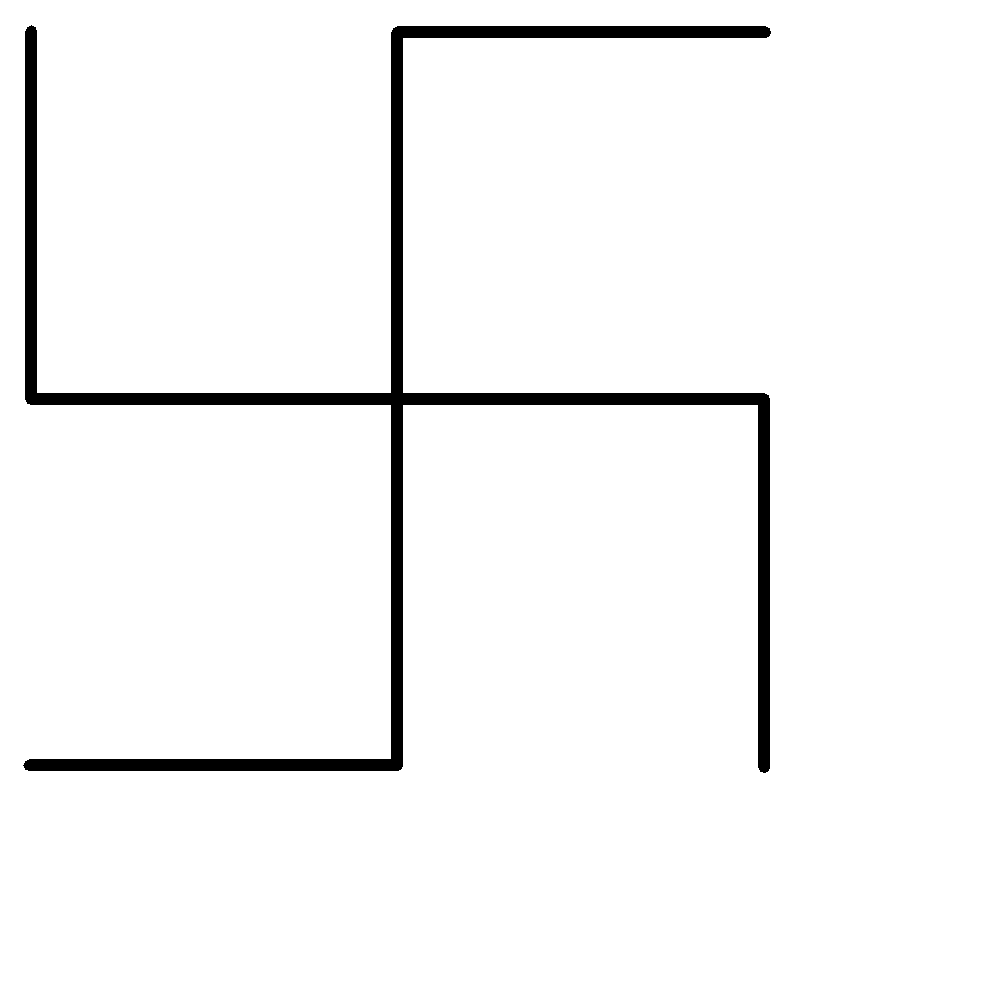
A.Neononane
B.Tetraethyl carbon
C.2-ethylpentane
D.3,3-diethyl pentane

Answer
624.9k+ views
Hint:
While naming hydrocarbons, look for the longest chain first. The longest chain in the hydrocarbon is the parent chain.
Complete step by step answer:
First of all, let us simplify the structure of the hydrocarbon given above:
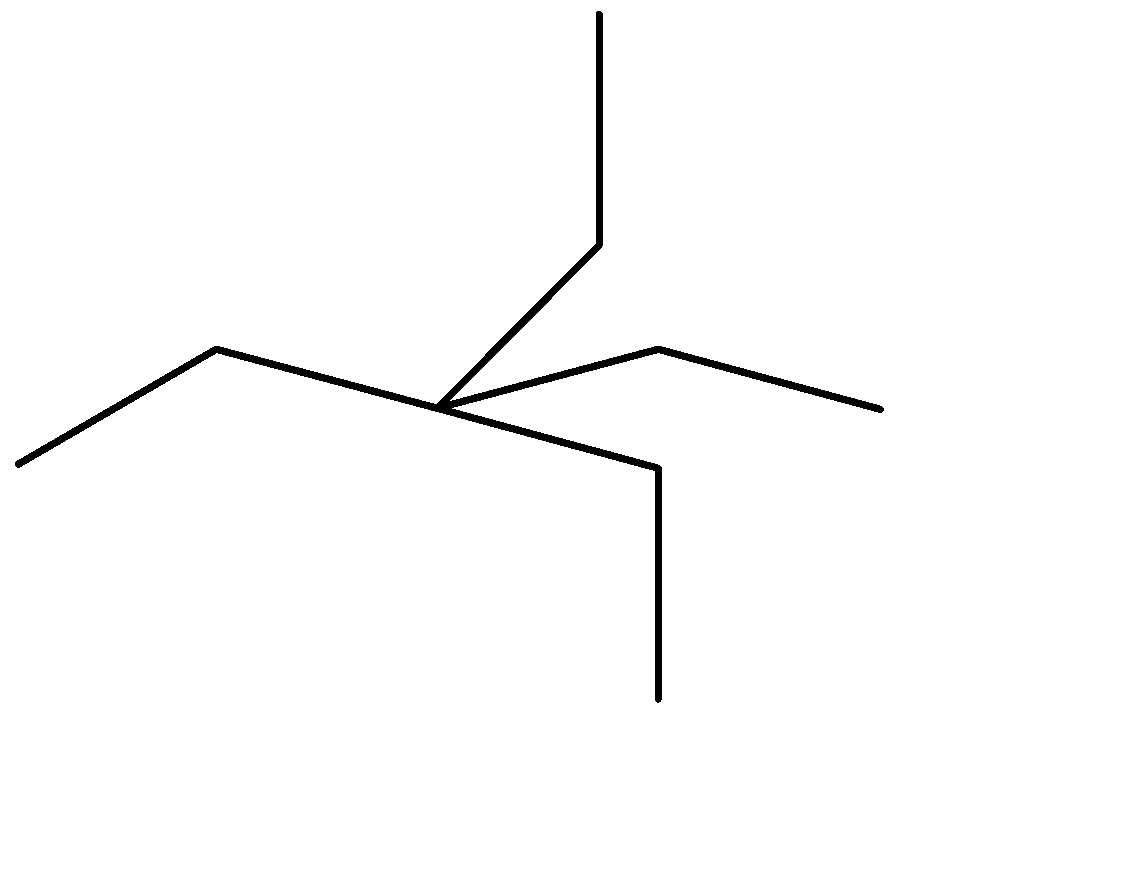
Simplifying it further, we get
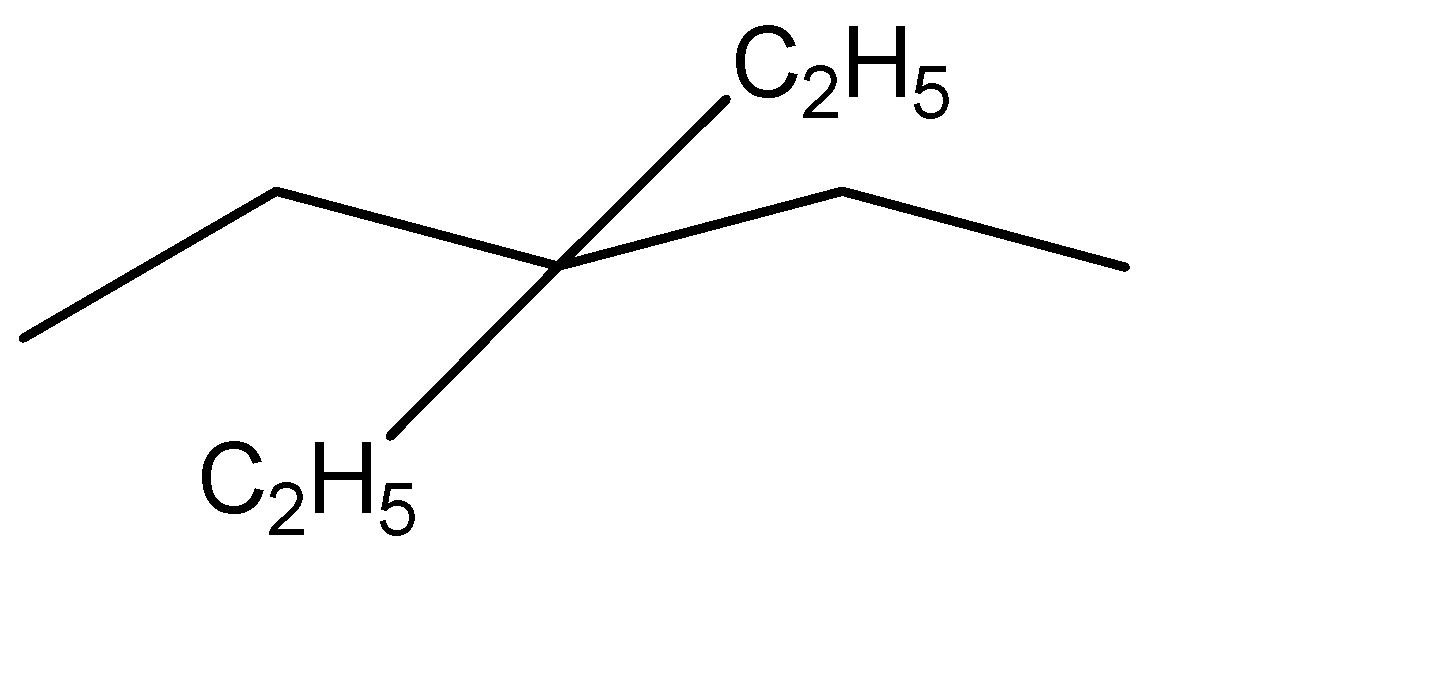
Now that we have the simplified representation of the given hydrocarbon, we can observe:
-The longest single carbon-carbon chain contains 5 carbon atoms.
-There are no double or triple carbon bonds on the longest chain, hence the given organic compound is an alkane.
-The only functional groups to be present on the longest chain are 2 ethyl groups on the third carbon atom from either side.
Now, let us discuss some rules for the nomenclature of such ‘alkanes’
-Find the longest chain of carbon atoms. This will be the parent chain of the compound.
Then identify all the substituents of the functional groups.
-While numbering the functional group, the numbering on the parent chain should be such that it should be closest to the terminal carbon atom.
-If the same functional group is occurring more than once, then while mentioning the functional group in the name of the hydrocarbon, appropriate position numbering should be mentioned. In addition, the number of times the substituent group occurs is indicated by a prefix (di, tri, tetra, etc.).
-If chains of equal length are competing for selection as the parent chain, then the choice goes in series to:
a) the chain which has the greatest number of side chains.
b) the chain whose substituents have the lowest- numbers.
c) the chain having the greatest number of carbon atoms in the smaller side chain.
d)the chain having the least branched side chains.
-Following these rules and observing the hydrocarbon in question, we can say that,
Parent chain has 5 carbon atoms and is hence named as pentane
2 Ethyl groups are present at the third position. His these will be named as 3,3-diethyl
Hence, the IUPAC nomenclature of the given compound is 3,3-diethyl pentane.
Hence, Option D is the correct option.
Note:
The names of the substituents formed by the removal of one hydrogen from the end of the chain is obtained by changing the suffix -ane to -yl.
While naming hydrocarbons, look for the longest chain first. The longest chain in the hydrocarbon is the parent chain.
Complete step by step answer:
First of all, let us simplify the structure of the hydrocarbon given above:

Simplifying it further, we get

Now that we have the simplified representation of the given hydrocarbon, we can observe:
-The longest single carbon-carbon chain contains 5 carbon atoms.
-There are no double or triple carbon bonds on the longest chain, hence the given organic compound is an alkane.
-The only functional groups to be present on the longest chain are 2 ethyl groups on the third carbon atom from either side.
Now, let us discuss some rules for the nomenclature of such ‘alkanes’
-Find the longest chain of carbon atoms. This will be the parent chain of the compound.
Then identify all the substituents of the functional groups.
-While numbering the functional group, the numbering on the parent chain should be such that it should be closest to the terminal carbon atom.
-If the same functional group is occurring more than once, then while mentioning the functional group in the name of the hydrocarbon, appropriate position numbering should be mentioned. In addition, the number of times the substituent group occurs is indicated by a prefix (di, tri, tetra, etc.).
-If chains of equal length are competing for selection as the parent chain, then the choice goes in series to:
a) the chain which has the greatest number of side chains.
b) the chain whose substituents have the lowest- numbers.
c) the chain having the greatest number of carbon atoms in the smaller side chain.
d)the chain having the least branched side chains.
-Following these rules and observing the hydrocarbon in question, we can say that,
Parent chain has 5 carbon atoms and is hence named as pentane
2 Ethyl groups are present at the third position. His these will be named as 3,3-diethyl
Hence, the IUPAC nomenclature of the given compound is 3,3-diethyl pentane.
Hence, Option D is the correct option.
Note:
The names of the substituents formed by the removal of one hydrogen from the end of the chain is obtained by changing the suffix -ane to -yl.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




