The hybridization of $P$ in $POC{l_3}$ and shape of $POC{l_3}$ are, respectively
A.$s{p^3}$, tetrahedral
B.$s{p^3}$, pyramidal
C.$s{p^3}$, square planar
D.$s{p^3}$, distorted tetrahedral
Answer
556.1k+ views
Hint: At first you should know about hybridization and the process to draw the structures of compounds. Hybridization is defined as the concept of mixing two atomic orbitals with the same energy levels to give new degenerate orbitals.
Complete answer:
In order to determine the hybridization, we need to draw the structure of $POC{l_3}$. To form a bond between the two atoms there should be sharing of electron pairs between the atoms. As we all know that chlorine has one lone pair and oxygen has two lone pairs of atoms, the phosphorus atom forms three single bonds with three chlorine atoms and a double with one oxygen atom.
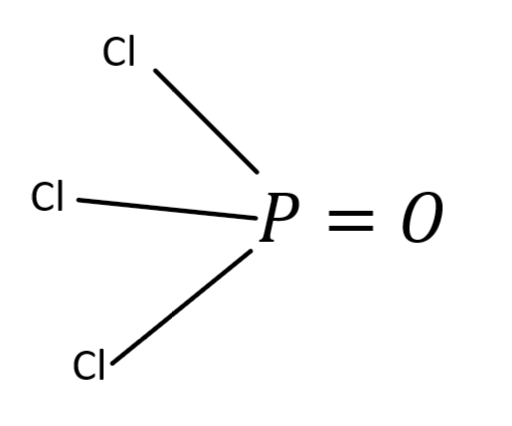
From the above diagram, we can say that P has $3\sigma $ bonds with three chlorine atoms and it formed a double bond with an oxygen atom. We all know that a double bond contains one $\sigma $ and one $\pi $ bond.
Hybridization of an atom can also be calculated by using no of $\sigma $bonds that the atom is attached to. So from the above diagram the P atom is attached to $4{\sigma _{}}$ bonds then the hybridization of P atom is $s{p^3}$.
The shape of $POC{l_3}$is tetrahedral due to the asymmetric charge distribution around the phosphorus atom.
So the answer is A.
Note:
To draw the structures of different compounds you should keep in mind about the lone pairs of atoms and its electronic configuration. Without knowing about the electronic configuration you cannot have a basic idea of valence electrons.
Complete answer:
In order to determine the hybridization, we need to draw the structure of $POC{l_3}$. To form a bond between the two atoms there should be sharing of electron pairs between the atoms. As we all know that chlorine has one lone pair and oxygen has two lone pairs of atoms, the phosphorus atom forms three single bonds with three chlorine atoms and a double with one oxygen atom.

From the above diagram, we can say that P has $3\sigma $ bonds with three chlorine atoms and it formed a double bond with an oxygen atom. We all know that a double bond contains one $\sigma $ and one $\pi $ bond.
Hybridization of an atom can also be calculated by using no of $\sigma $bonds that the atom is attached to. So from the above diagram the P atom is attached to $4{\sigma _{}}$ bonds then the hybridization of P atom is $s{p^3}$.
The shape of $POC{l_3}$is tetrahedral due to the asymmetric charge distribution around the phosphorus atom.
So the answer is A.
Note:
To draw the structures of different compounds you should keep in mind about the lone pairs of atoms and its electronic configuration. Without knowing about the electronic configuration you cannot have a basic idea of valence electrons.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




