The formation of alkene from alkyl halide is an example of
A. Addition
B. Elimination
C. Substitution
D. Option a and c
Answer
381.6k+ views
Hint: The alkyl halide is a compound containing a hydrocarbon chain and halide group present in the chain such as RX where R is an alkyl group or long chain of hydrocarbons and X is a halogen. Now alkyl halide alkene can be obtained when the halogen group from alpha carbon (carbon bonded to halogen group) and hydrogen from beta carbon are removed so that both alpha and beta carbon form a pi bond resulting in the formation of a double bond (alkene).
Complete Step by Step Answer:
Alkyl halide (ethyl halide) when made react with aq. KOH then potassium hydroxide behave as a strong nucleophile and \[O{{H}^{-}}\]attach on carbocation which is formed as halogen of alkyl halide leave the chain taking away the bond pair (from alpha hydrogen). In this reaction \[O{{H}^{-}}\] is substituted in the place of Br so we can say this is a substitution reaction such as
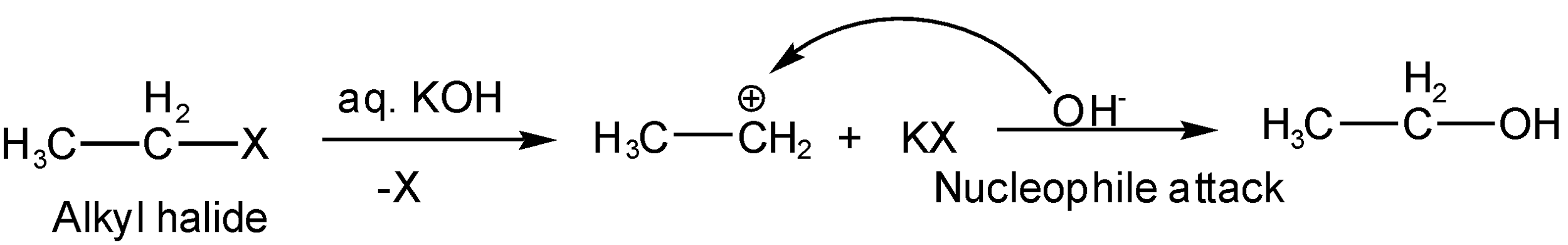
If the alkyl halide (ethyl halide) is made to react with alcoholic KOH then potassium hydroxide behaves like a strong base (means forming a bond with electron-deficient species mostly hydrogen ions). The strong base release \[O{{H}^{-}}\] and \[{{K}^{+}}\]ions in the presence of alkyl halide. Then \[{{K}^{+}}\]attack on halide of alkyl halide creating carbocation (Br removed from alpha hydrogen) but \[O{{H}^{-}}\] will not attack carbocation but take away hydrogen ion (\[{{H}^{+}}\]) from beta carbon which results in the formation of pi bond and thus double bond (alkene). In this reaction, hydrogen is eliminated so this is an elimination reaction such as
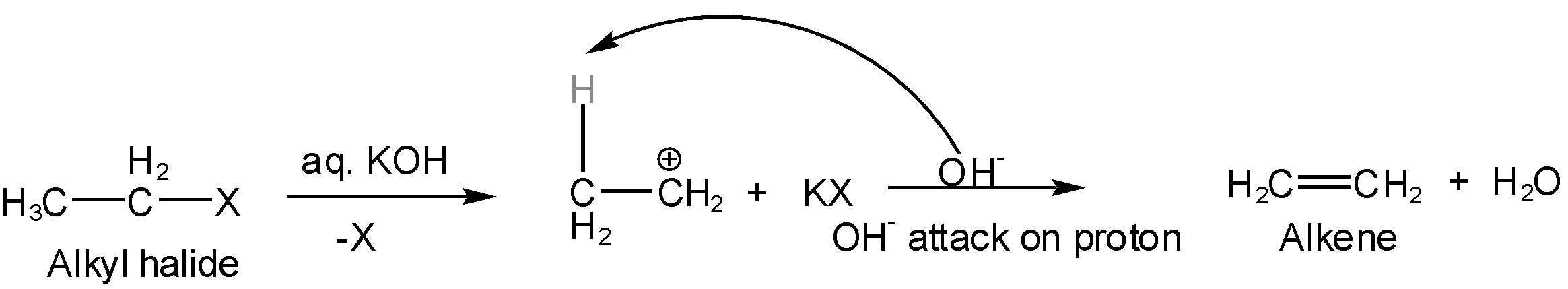
Thus, the correct option is B.
Note: The addition reaction cannot be used to form alkene from alkyl halide as it is used to from alkane, saturated hydrocarbons from alkene or alkynes, unsaturated hydrocarbons such as
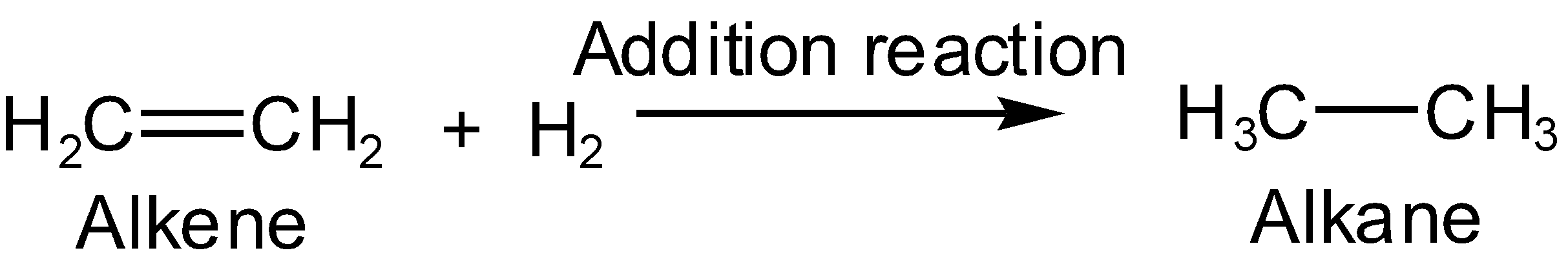
In the above reaction, both hydrogens are added to the ethene (alkene) to give ethane (alkane).
Complete Step by Step Answer:
Alkyl halide (ethyl halide) when made react with aq. KOH then potassium hydroxide behave as a strong nucleophile and \[O{{H}^{-}}\]attach on carbocation which is formed as halogen of alkyl halide leave the chain taking away the bond pair (from alpha hydrogen). In this reaction \[O{{H}^{-}}\] is substituted in the place of Br so we can say this is a substitution reaction such as

If the alkyl halide (ethyl halide) is made to react with alcoholic KOH then potassium hydroxide behaves like a strong base (means forming a bond with electron-deficient species mostly hydrogen ions). The strong base release \[O{{H}^{-}}\] and \[{{K}^{+}}\]ions in the presence of alkyl halide. Then \[{{K}^{+}}\]attack on halide of alkyl halide creating carbocation (Br removed from alpha hydrogen) but \[O{{H}^{-}}\] will not attack carbocation but take away hydrogen ion (\[{{H}^{+}}\]) from beta carbon which results in the formation of pi bond and thus double bond (alkene). In this reaction, hydrogen is eliminated so this is an elimination reaction such as

Thus, the correct option is B.
Note: The addition reaction cannot be used to form alkene from alkyl halide as it is used to from alkane, saturated hydrocarbons from alkene or alkynes, unsaturated hydrocarbons such as
In the above reaction, both hydrogens are added to the ethene (alkene) to give ethane (alkane).
Recently Updated Pages
Master Class 10 Computer Science: Engaging Questions & Answers for Success

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Master Class 10 Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Explain the Treaty of Vienna of 1815 class 10 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Find the sum of series 1 + 2 + 3 + 4 + 5 + + 100 class 9 maths CBSE




