Sodium benzene sulphonate after reaction with $NaOH$ undergoes acidic hydrolysis, the compound formed would be:
(a)- Phenol
(b)- Benzoic acid
(c)- Benzene
(d)- Disodium benzaldehyde
Answer
594.6k+ views
Hint: Sodium benzene sulphonate is an aromatic compound in which the carbon atom of the benzene is attached to $-S{{O}_{3}}Na$ group. When the sodium benzene sulphonate is treated with sodium hydroxide, the by-products are sodium sulfite and water.
Complete Solution :
Sodium benzene sulphonate is an aromatic compound in which the carbon atom of the benzene is attached to $-S{{O}_{3}}Na$ group. The structure of sodium benzene sulphonate is given below:
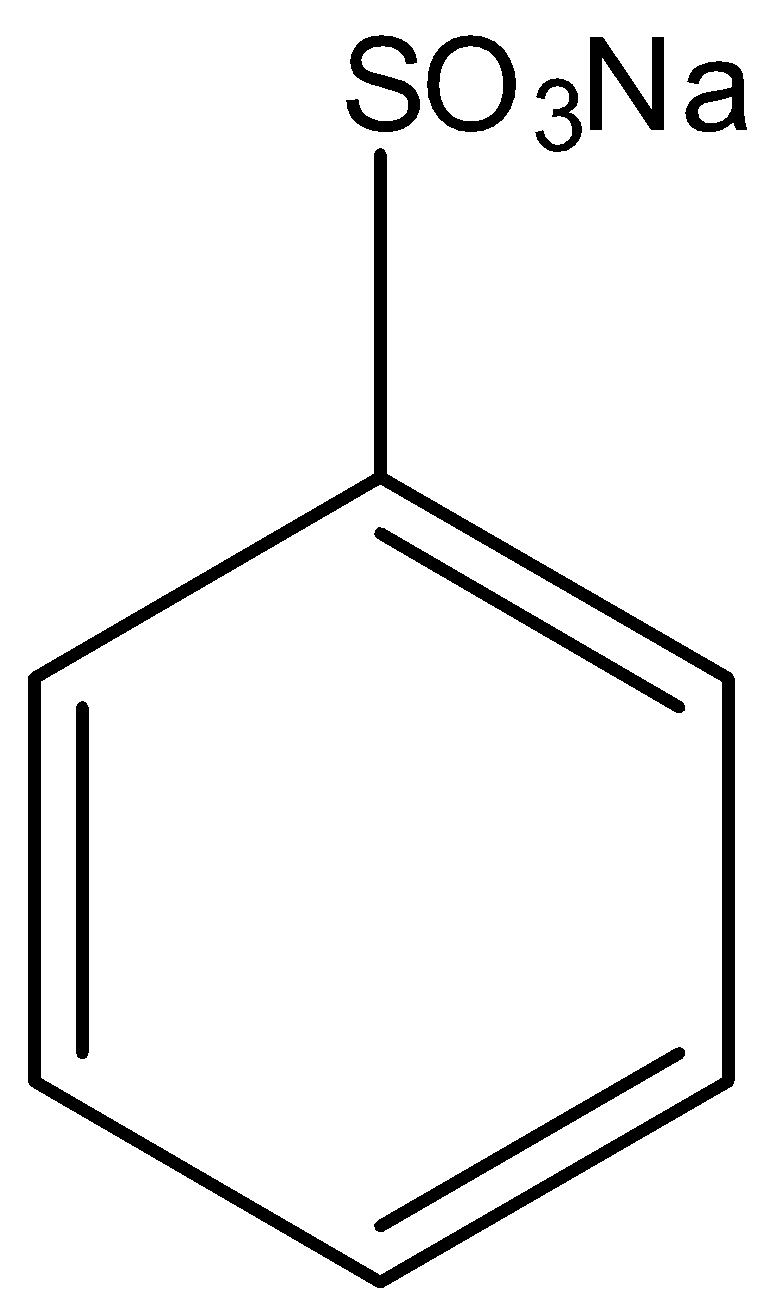
When the sodium benzene sulphonate is reacted with sodium hydroxide, the sodium sulfonate group from the benzene is replaced with the sodium oxide group, and there is the formation of sodium phenoxide and the by-products are sodium sulfite and water. Now when this sodium phenoxide is hydrolyzed means the sodium phenoxide is reacted with water, the sodium ion in the sodium phenoxide is replaced with a hydrogen ion, so there is formation of phenol. And the by-product of this reaction is sodium hydroxide. This reaction is the acidification of sodium phenoxide forming phenol. This reaction can only occur in the presence of acid. The two-step reaction is given below:
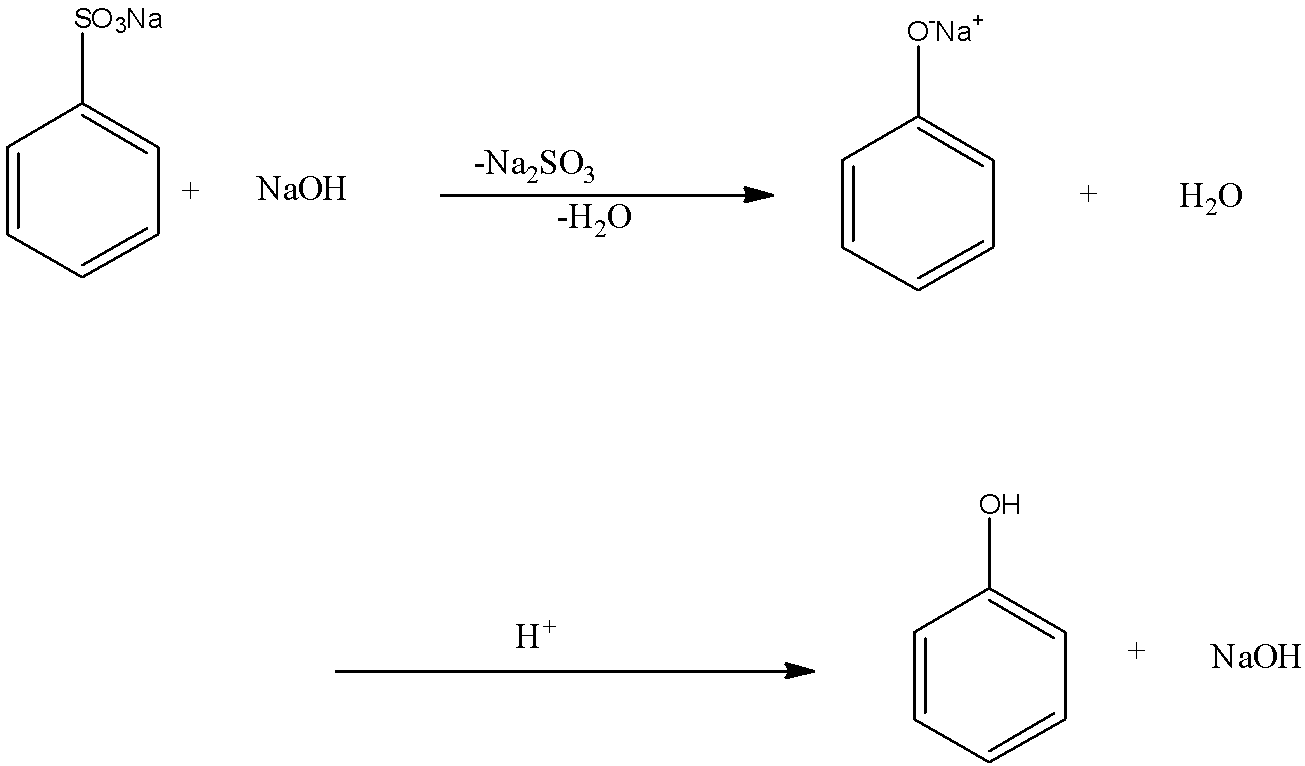
So the final product in this reaction is phenol. Therefore, the correct answer is an option (a)- Phenol.
So, the correct answer is “Option A”.
Note: This is one main process for the production of phenol and this process is mostly followed in the laboratory. The temperature for this reaction is 573 – 623 K. The sodium benzene sulphonate can be easily produced from benzene sulphonic acid.
Complete Solution :
Sodium benzene sulphonate is an aromatic compound in which the carbon atom of the benzene is attached to $-S{{O}_{3}}Na$ group. The structure of sodium benzene sulphonate is given below:

When the sodium benzene sulphonate is reacted with sodium hydroxide, the sodium sulfonate group from the benzene is replaced with the sodium oxide group, and there is the formation of sodium phenoxide and the by-products are sodium sulfite and water. Now when this sodium phenoxide is hydrolyzed means the sodium phenoxide is reacted with water, the sodium ion in the sodium phenoxide is replaced with a hydrogen ion, so there is formation of phenol. And the by-product of this reaction is sodium hydroxide. This reaction is the acidification of sodium phenoxide forming phenol. This reaction can only occur in the presence of acid. The two-step reaction is given below:

So the final product in this reaction is phenol. Therefore, the correct answer is an option (a)- Phenol.
So, the correct answer is “Option A”.
Note: This is one main process for the production of phenol and this process is mostly followed in the laboratory. The temperature for this reaction is 573 – 623 K. The sodium benzene sulphonate can be easily produced from benzene sulphonic acid.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




