Show how could you accomplish the following synthesis?
Cyclohexyl amine $\longrightarrow$ N- cyclohexyl acetamide
Answer
622.8k+ views
Hint: Cyclohexylamine is a cyclic, aromatic structure with 6 carbon and an amine group attached to it. For the conversion of amine to the amide group, the hydrogen is removed from the amine and carbonyl group (\[\text{C=O}\]) attaches in place of hydrogen and forms acetamide group i.e. \[\text{N}{{\text{H}}_{3}}\text{-CO-N}{{\text{H}}_{3}}\text{ }\] .
Complete answer:
-To convert the cyclohexylamine let's draw the structure first.
-As it has 6- membered cyclic ring with an amine group so the structure is:
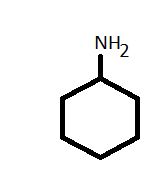
-Now, in N - cyclohexyl acetamide has a similar structure to cyclohexylamine except the presence of acetamide group in place of the amine group.
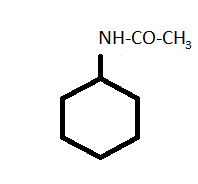
-For the synthesis of N - cyclohexyl acetamide from cyclohexylamine, pyridine is used as a catalyst to increase the rate or speed of the reaction.
-Along with the pyridine, acetyl chloride is used which has a molecular formula of $\text{C}{{\text{H}}_{3}}\text{-CO-Cl}$.
-From this, the oxidation of cyclohexylamine takes place due to which hydrogen atom releases from $\text{-N}{{\text{H}}_{2}}$ the group.
-Meanwhile, from acetyl chloride chlorine is released.
-So, the \[\text{C}{{\text{H}}_{3}}\text{ - CO-}\] group attaches with the amine group of the cyclohexylamine and it forms cyclohexyl acetamide.
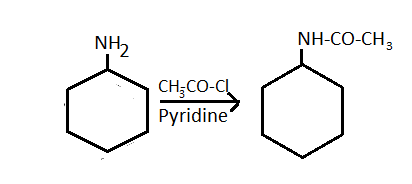
Cyclohexylamine N - cyclohexyl acetamide
-Along with it, hydrochloric acid is also formed.
-So, by using acetyl chloride we can synthesize N - cyclohexyl acetamide.
Note: It is observed that when N - cyclohexyl acetamide is reduced by $\text{LiAl}{{\text{H}}_{4}}$ which is a strong reducing agent, then the oxygen is lost from the acetamide and it can form N - ethyl cyclohexylamine, which has extra alkyl group than cyclohexylamine.
.
Complete answer:
-To convert the cyclohexylamine let's draw the structure first.
-As it has 6- membered cyclic ring with an amine group so the structure is:

-Now, in N - cyclohexyl acetamide has a similar structure to cyclohexylamine except the presence of acetamide group in place of the amine group.

-For the synthesis of N - cyclohexyl acetamide from cyclohexylamine, pyridine is used as a catalyst to increase the rate or speed of the reaction.
-Along with the pyridine, acetyl chloride is used which has a molecular formula of $\text{C}{{\text{H}}_{3}}\text{-CO-Cl}$.
-From this, the oxidation of cyclohexylamine takes place due to which hydrogen atom releases from $\text{-N}{{\text{H}}_{2}}$ the group.
-Meanwhile, from acetyl chloride chlorine is released.
-So, the \[\text{C}{{\text{H}}_{3}}\text{ - CO-}\] group attaches with the amine group of the cyclohexylamine and it forms cyclohexyl acetamide.

Cyclohexylamine N - cyclohexyl acetamide
-Along with it, hydrochloric acid is also formed.
-So, by using acetyl chloride we can synthesize N - cyclohexyl acetamide.
Note: It is observed that when N - cyclohexyl acetamide is reduced by $\text{LiAl}{{\text{H}}_{4}}$ which is a strong reducing agent, then the oxygen is lost from the acetamide and it can form N - ethyl cyclohexylamine, which has extra alkyl group than cyclohexylamine.
.
Recently Updated Pages
Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Master Class 9 Social Science: Engaging Questions & Answers for Success

Class 9 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




