P-P bond is present in:
(A) ${{P}_{4}}{{O}_{10}}$
(B) ${{P}_{4}}{{O}_{6}}$
(C) ${{H}_{4}}{{P}_{2}}{{O}_{6}}$
(D) ${{H}_{4}}{{P}_{2}}{{O}_{7}}$
Answer
600.6k+ views
Hint: P-P bond means that the two phosphorous atoms are joined with each other which leads to formation of a bridge between the molecules. P-P is a covalent bond which is formed by the sharing of electrons by different atoms which are present in the molecule.
Complete step by step solution:
Hypophosphoric acid i.e. ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ , in this hypo means beneath or less than. The Hypophosphoric acid has an oxidation state of +4 between the phosphorus acid and the phosphoric acid.
The oxidation state of P in${{P}_{4}}{{O}_{10}}$, ${{P}_{4}}{{O}_{6}}$ and ${{H}_{4}}{{P}_{2}}{{O}_{7}}$ is +5. The calculated oxidation state is equal to the maximum oxidation state of phosphorus hence there is P-O-P bond present in all these compounds.
Among the given options Hypophosphoric acid i.e. ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ forms a P-P double bond because the oxidation state if +4 which is less than the maximum oxidation state of P. and when the calculated oxidation state is less than the maximum oxidation state then there is a direct linkage between the elements here it is P-P linkage.
The structure of Hypophosphoric acid i.e. ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ is shown below:
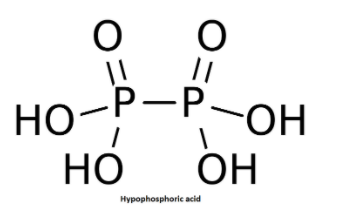
In this structure we can note that there are two P-O bonds having bond length of 151 pm and four P-OH bonds having bond length of 151 pm and one P-P single bond having bond length of 219 pm.
Hence the correct answer is option ©
Note: Hypophosphoric acid i.e. ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ is used as a bleaching agent. It is also used as a reducing agent, as a stimulant and pharmaceutical agent and also as a wetting agent. Hypophosphoric acid is a mineral acid which is present as dehydrate in the solid state.
Complete step by step solution:
Hypophosphoric acid i.e. ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ , in this hypo means beneath or less than. The Hypophosphoric acid has an oxidation state of +4 between the phosphorus acid and the phosphoric acid.
The oxidation state of P in${{P}_{4}}{{O}_{10}}$, ${{P}_{4}}{{O}_{6}}$ and ${{H}_{4}}{{P}_{2}}{{O}_{7}}$ is +5. The calculated oxidation state is equal to the maximum oxidation state of phosphorus hence there is P-O-P bond present in all these compounds.
Among the given options Hypophosphoric acid i.e. ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ forms a P-P double bond because the oxidation state if +4 which is less than the maximum oxidation state of P. and when the calculated oxidation state is less than the maximum oxidation state then there is a direct linkage between the elements here it is P-P linkage.
The structure of Hypophosphoric acid i.e. ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ is shown below:

In this structure we can note that there are two P-O bonds having bond length of 151 pm and four P-OH bonds having bond length of 151 pm and one P-P single bond having bond length of 219 pm.
Hence the correct answer is option ©
Note: Hypophosphoric acid i.e. ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ is used as a bleaching agent. It is also used as a reducing agent, as a stimulant and pharmaceutical agent and also as a wetting agent. Hypophosphoric acid is a mineral acid which is present as dehydrate in the solid state.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
How many of the following diseases can be studied with class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Receptor sites for neurotransmitters are present on class 11 biology CBSE

JGA JuxtaGlomerular Apparatus a sensitive region which class 11 biology CBSE

Root hairs develop from the region of A Meristematic class 11 biology CBSE

The alphahelix and betasheet are found at which level class 11 biology CBSE




