p-Nitrophenol is a stronger acid than phenol while p-cresol is a weaker acid. This can be explained as:
A.${\text{ - C}}{{\text{H}}_{\text{3}}}$ group decreases the electron density on the oxygen of O-H group making p-cresol a weaker acid.
B.${\text{ - N}}{{\text{O}}_{\text{2}}}$ decreases the electron density on the oxygen of O-H group making p-nitrophenol a stronger acid.
C.${\text{ - C}}{{\text{H}}_{\text{3}}}$ group increases the electron density on the oxygen of O-H group making the release of ${{\text{H}}^{\text{ + }}}$ easier
D.${\text{ - N}}{{\text{O}}_{\text{2}}}$ group increases the electron density on oxygen of O-H group making release of ${{\text{H}}^{\text{ + }}}$easier
Answer
607.8k+ views
Hint:The p-nitrophenol is a nitro group attached to the para position of phenol. Similarly, p-cresol is a methyl group attached to the para position of phenol. An electronegative atom pulls the electrons and has high electron density. The presence of the electron-withdrawing group and electron-donating group affects the acidity of the compounds. Acidity is determined by the ability to release ${{\text{H}}^{\text{ + }}}$ ions.
Complete step by step answer:
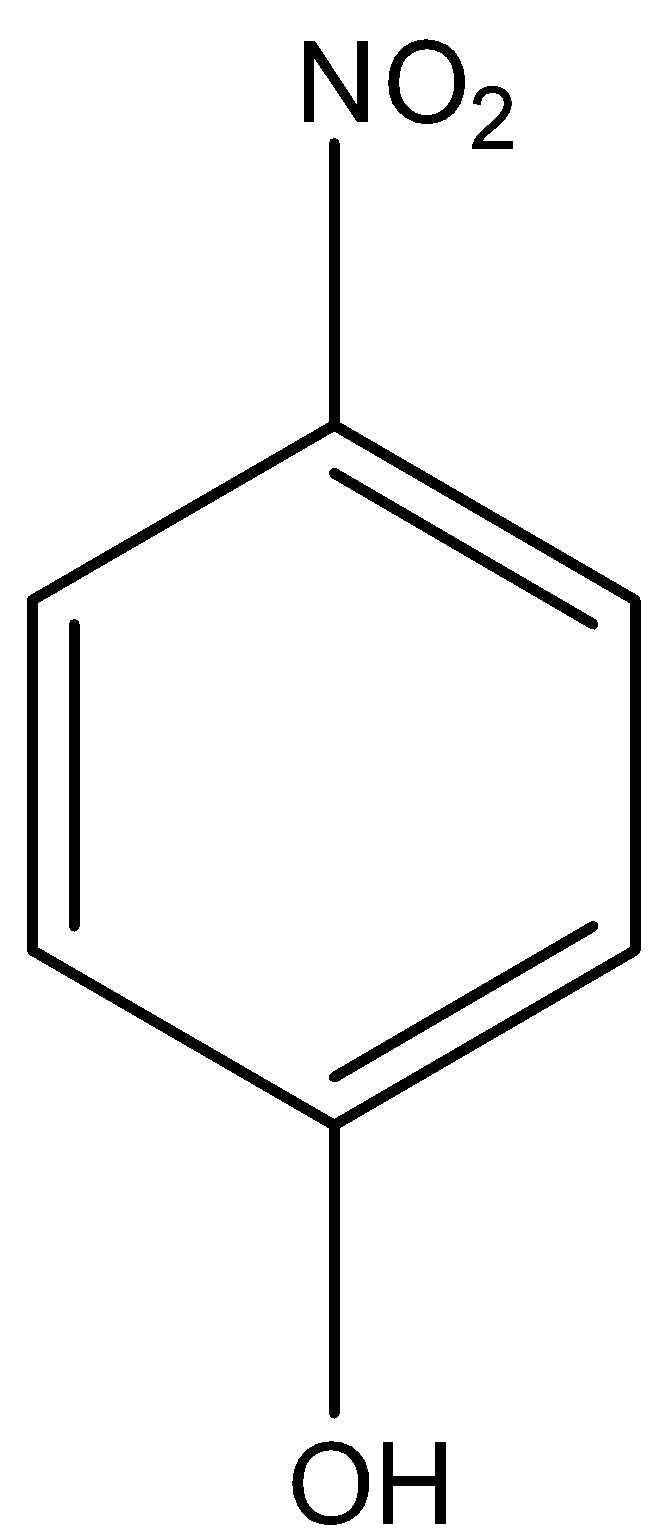
Let us look at the structure of p-nitrophenol. Phenol is an electron-deficient group.
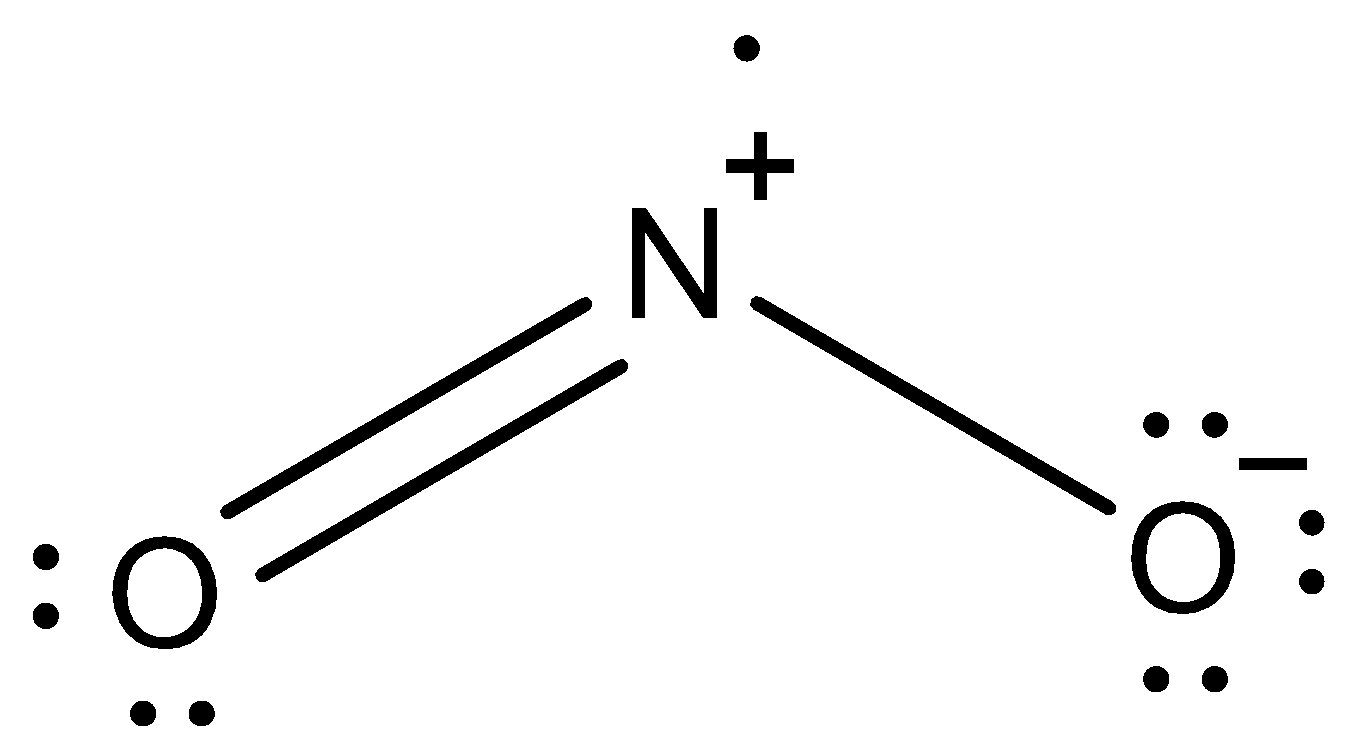
This is the lewis dot structure of ${\text{N}}{{\text{O}}_{\text{2}}}$.
We can see that Nitrogen in ${\text{N}}{{\text{O}}_{\text{2}}}$ already has a positive charge on it. It is because it gave one of its lone pairs to oxygen (double bond). When this nitrogen is attached to the benzene ring, the nitrogen will pull the electrons from the benzene ring to reduce the positive charge.
Therefore, ${\text{N}}{{\text{O}}_{\text{2}}}$ is an electron-withdrawing group.
In p-nitrophenol, since it is ${\text{N}}{{\text{O}}_{\text{2}}}$ is an electron-withdrawing group, all the electron density will be drifted towards ${\text{N}}{{\text{O}}_{\text{2}}}$
This will result in a decrease in the electron density of oxygen in the O-H bond.
Thus, the release of ${{\text{H}}^{\text{ + }}}$ is easier.
Now, let us look at o-cresol.
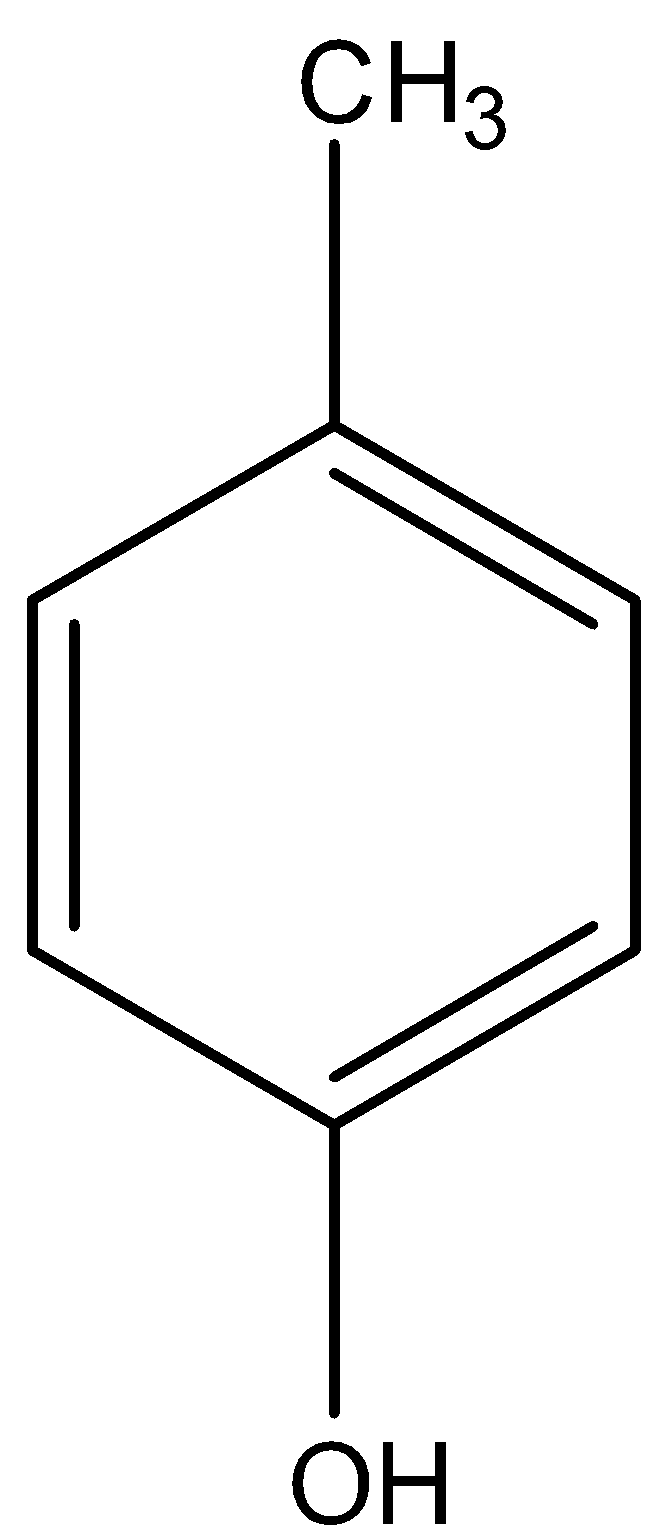
The ${\text{ - C}}{{\text{H}}_{\text{3}}}$ is attached to the para position of phenol.
We know that the methyl group in cresol is electron-donating. It gives its electron density to the electron-deficient phenol. Thus, the electron density on the O-H group increases.
Therefore, it cannot give out ${{\text{H}}^{\text{ + }}}$ ions.
Therefore, the correct answer is that the ${\text{ - N}}{{\text{O}}_{\text{2}}}$ decreases the electron density on the oxygen of the O-H group making p-nitrophenol a stronger acid.
Thus, the correct option is (B).
Note: The presence of an electron-donating group will decrease the acidity. This is because the electron-donating group increases the electron density on the electron-deficient species. Thus, it makes it difficult to release ${{\text{H}}^{\text{ + }}}$ ions as the bonds are stronger. The presence of electron-withdrawing groups ${\text{ - N}}{{\text{O}}_{\text{2}}}$ decreases the electron density and facilitates the release of ${{\text{H}}^{\text{ + }}}$ ions. Other examples of electron-donating groups are ${\text{ - N}}{{\text{H}}_{\text{2}}}$, -NR, - etc., and the electron-withdrawing groups are halogen, ${\text{ - N}}{{\text{O}}_{\text{2}}}$, ${\text{ - C}}{{\text{F}}_{\text{3}}}$
Complete step by step answer:
Let us look at the structure of p-nitrophenol. Phenol is an electron-deficient group.


This is the lewis dot structure of ${\text{N}}{{\text{O}}_{\text{2}}}$.
We can see that Nitrogen in ${\text{N}}{{\text{O}}_{\text{2}}}$ already has a positive charge on it. It is because it gave one of its lone pairs to oxygen (double bond). When this nitrogen is attached to the benzene ring, the nitrogen will pull the electrons from the benzene ring to reduce the positive charge.
Therefore, ${\text{N}}{{\text{O}}_{\text{2}}}$ is an electron-withdrawing group.
In p-nitrophenol, since it is ${\text{N}}{{\text{O}}_{\text{2}}}$ is an electron-withdrawing group, all the electron density will be drifted towards ${\text{N}}{{\text{O}}_{\text{2}}}$
This will result in a decrease in the electron density of oxygen in the O-H bond.
Thus, the release of ${{\text{H}}^{\text{ + }}}$ is easier.
Now, let us look at o-cresol.

The ${\text{ - C}}{{\text{H}}_{\text{3}}}$ is attached to the para position of phenol.
We know that the methyl group in cresol is electron-donating. It gives its electron density to the electron-deficient phenol. Thus, the electron density on the O-H group increases.
Therefore, it cannot give out ${{\text{H}}^{\text{ + }}}$ ions.
Therefore, the correct answer is that the ${\text{ - N}}{{\text{O}}_{\text{2}}}$ decreases the electron density on the oxygen of the O-H group making p-nitrophenol a stronger acid.
Thus, the correct option is (B).
Note: The presence of an electron-donating group will decrease the acidity. This is because the electron-donating group increases the electron density on the electron-deficient species. Thus, it makes it difficult to release ${{\text{H}}^{\text{ + }}}$ ions as the bonds are stronger. The presence of electron-withdrawing groups ${\text{ - N}}{{\text{O}}_{\text{2}}}$ decreases the electron density and facilitates the release of ${{\text{H}}^{\text{ + }}}$ ions. Other examples of electron-donating groups are ${\text{ - N}}{{\text{H}}_{\text{2}}}$, -NR, - etc., and the electron-withdrawing groups are halogen, ${\text{ - N}}{{\text{O}}_{\text{2}}}$, ${\text{ - C}}{{\text{F}}_{\text{3}}}$
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




