Perxenate ion is:
A.\[Xe{O_6}^{4 - }\]
B.\[HXe{O_4}^ - \]
C.\[Xe{O_4}^{2 - }\]
D.\[Xe{O_4}^ - \]
Answer
615.3k+ views
Hint: Perxenate belongs to compounds of xenon having the valency of xenon more than eight. They are also referred to as hypervalent compounds of xenon. They exhibit octahedral geometry.
Complete step by step answer:
Step by step solution:
Perxenates are oxides of xenon compounds. They are the anions having octahedral geometry. They are yellow in colour. The \[Xe - O\] bond length is \[1.875{\text{ }}{A}\] as measured using X-ray crystallography. The bond angles of \[O - Xe - O\] vary in the range of \[87^\circ \] to \[93^\circ \]. The molecular formula of perxenate ion is \[Xe{O_6}^{4 - }\] .
The correct answer is A i.e. \[Xe{O_6}^{4 - }\]. The structure of perxenate ion can be shown as:
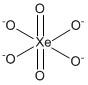
The valence electrons of \[Xe\] are eight so the covalency of \[Xe\] is eight. The number of hybrid orbitals is equal to six (six bond pairs + zero lone pairs). Therefore, the hybridization of \[Xe{O_6}^{4 - }\] is \[s{p^3}{d^2}\]. As seen in the structure two oxygen atoms form double and four oxygen atoms form single for fulfilling the octet.
The perxenate ion is produced by treating xenon trioxide with strong alkali.
$2Xe{O_3}(s) + 4O{H^ - }\xrightarrow{{}}Xe(g) + Xe{O_6}^{4 - }(aq) + {O_2}(g) + 2{H_2}O(l)$
Perxenate ions are strong oxidizing agents and can oxidize a variety of metals like silver, copper and manganese. They are reduced to \[HXe{O_4}^ - \] in presence of acids.
\[HXe{O_4}^ - \] are known as xenates. They are prepared by the reaction of xenon trioxide with aqueous hydroxides. Actually they are the intermediates during the formation of perxenate. The xenon trioxide reacts with hydroxide ion to give xenates which on further reaction with hydroxide ion to yield perxenate.
$Xe{O_3}(aq) + 2O{H^ - }(aq)\xrightarrow{{}}2HXe{O_4}^ - (aq)$
\[2HXe{O_4}^ - (aq) + 2O{H^ - }(aq)\xrightarrow{{}}Xe{O_6}^{4 - }(aq) + Xe(aq) + {O_2}(g) + 2{H_2}O(l)\]
\[Xe{O_4}\] is one of the existing oxides of xenon, but \[Xe{O_4}^{2 - }\] and \[Xe{O_4}^ - \] do not exist.
Note: Perxenates should not be confused with peroxides of xenon. Perxenate does not contain any peroxide linkages. Perxenates only have single and double bonds with oxygen atoms.
Complete step by step answer:
Step by step solution:
Perxenates are oxides of xenon compounds. They are the anions having octahedral geometry. They are yellow in colour. The \[Xe - O\] bond length is \[1.875{\text{ }}{A}\] as measured using X-ray crystallography. The bond angles of \[O - Xe - O\] vary in the range of \[87^\circ \] to \[93^\circ \]. The molecular formula of perxenate ion is \[Xe{O_6}^{4 - }\] .
The correct answer is A i.e. \[Xe{O_6}^{4 - }\]. The structure of perxenate ion can be shown as:

The valence electrons of \[Xe\] are eight so the covalency of \[Xe\] is eight. The number of hybrid orbitals is equal to six (six bond pairs + zero lone pairs). Therefore, the hybridization of \[Xe{O_6}^{4 - }\] is \[s{p^3}{d^2}\]. As seen in the structure two oxygen atoms form double and four oxygen atoms form single for fulfilling the octet.
The perxenate ion is produced by treating xenon trioxide with strong alkali.
$2Xe{O_3}(s) + 4O{H^ - }\xrightarrow{{}}Xe(g) + Xe{O_6}^{4 - }(aq) + {O_2}(g) + 2{H_2}O(l)$
Perxenate ions are strong oxidizing agents and can oxidize a variety of metals like silver, copper and manganese. They are reduced to \[HXe{O_4}^ - \] in presence of acids.
\[HXe{O_4}^ - \] are known as xenates. They are prepared by the reaction of xenon trioxide with aqueous hydroxides. Actually they are the intermediates during the formation of perxenate. The xenon trioxide reacts with hydroxide ion to give xenates which on further reaction with hydroxide ion to yield perxenate.
$Xe{O_3}(aq) + 2O{H^ - }(aq)\xrightarrow{{}}2HXe{O_4}^ - (aq)$
\[2HXe{O_4}^ - (aq) + 2O{H^ - }(aq)\xrightarrow{{}}Xe{O_6}^{4 - }(aq) + Xe(aq) + {O_2}(g) + 2{H_2}O(l)\]
\[Xe{O_4}\] is one of the existing oxides of xenon, but \[Xe{O_4}^{2 - }\] and \[Xe{O_4}^ - \] do not exist.
Note: Perxenates should not be confused with peroxides of xenon. Perxenate does not contain any peroxide linkages. Perxenates only have single and double bonds with oxygen atoms.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




