Oxidation of toluene to benzaldehyde by the use of chromyl chloride is called?
A) Wurtz reaction
B) Etard’s reaction
C) Fittig reaction
D) Rosenmund’s reaction
Answer
598.2k+ views
Hint:We know that benzaldehyde is an organic compound which consists of a benzene ring to which a formyl $\left( { - {\text{CHO}}} \right)$ group is attached. Chromyl chloride behaves as a weak acid in a nonpolar solvent. The methyl group of toluene gets partially oxidised by the chromyl chloride in this reaction.
Complete answer
The conversion of toluene to benzaldehyde by the use of chromyl chloride is an oxidation reaction. This reaction is known as Etard’s reaction. In this reaction, the compounds having at least one methyl group bonded to the benzene ring are required. The methyl group gets oxidised and converts to an aldehyde group. The oxidation of the methyl group is done using a weak oxidising agent. Thus, chromyl chloride which is a weak oxidising agent is used.
The overall mechanism of the reaction is as follows:
Step 1: The hydrogen from the methyl group of toluene is abstracted by the chromyl chloride. The chromyl chloride forms a bond with the benzene ring. This complex is known as Etard’s complex.
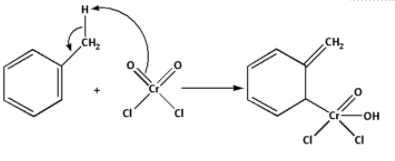
Step 2: The Etard’s complex formed in the first step undergoes rearrangement. The complex undergoes a 2,3-sigmatropic rearrangement. Thus, a rearrangement product is formed.
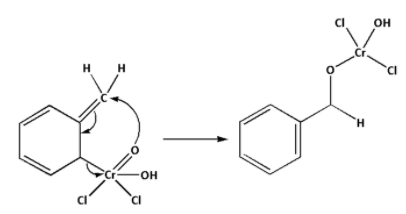
Step 3: The product formed in the above step is the rearrangement product. and are eliminated from the rearrangement product. This leads to the formation of benzaldehyde.
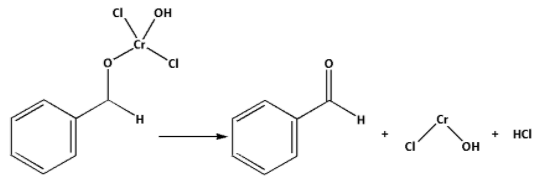
Thus, oxidation of toluene to benzaldehyde by the use of chromyl chloride is called Etard’s reaction.
Thus, the correct answer is option (B) Etard’s reaction.
Note:
i) Wurtz-Fittig reaction: In this reaction, two alkyl halides react with sodium metal in dry ether and produce a higher alkane.
ii) Rosenmund’s reaction: In this reaction, an acyl chloride is hydrogenated over catalyst, palladium on barium sulphate to produce aldehyde.
Complete answer
The conversion of toluene to benzaldehyde by the use of chromyl chloride is an oxidation reaction. This reaction is known as Etard’s reaction. In this reaction, the compounds having at least one methyl group bonded to the benzene ring are required. The methyl group gets oxidised and converts to an aldehyde group. The oxidation of the methyl group is done using a weak oxidising agent. Thus, chromyl chloride which is a weak oxidising agent is used.
The overall mechanism of the reaction is as follows:
Step 1: The hydrogen from the methyl group of toluene is abstracted by the chromyl chloride. The chromyl chloride forms a bond with the benzene ring. This complex is known as Etard’s complex.

Step 2: The Etard’s complex formed in the first step undergoes rearrangement. The complex undergoes a 2,3-sigmatropic rearrangement. Thus, a rearrangement product is formed.

Step 3: The product formed in the above step is the rearrangement product. and are eliminated from the rearrangement product. This leads to the formation of benzaldehyde.

Thus, oxidation of toluene to benzaldehyde by the use of chromyl chloride is called Etard’s reaction.
Thus, the correct answer is option (B) Etard’s reaction.
Note:
i) Wurtz-Fittig reaction: In this reaction, two alkyl halides react with sodium metal in dry ether and produce a higher alkane.
ii) Rosenmund’s reaction: In this reaction, an acyl chloride is hydrogenated over catalyst, palladium on barium sulphate to produce aldehyde.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE




