Osmoscope is used for
(a)Measuring TP (turgor pressure)
(b)Measuring OP (osmotic pressure)
(c)Measuring W (water potential)
(d)Demonstrating osmosis
Answer
615.6k+ views
Hint: It is a special type of process of movement of solvent from an area of a higher number to an area of lower number through a semipermeable membrane until the number of molecules on both sides is equal.
Complete answer:
Osmoscope is an instrument used for the measurement of osmosis. The instrument contains a beaker into which a semipermeable membrane. The inner of the semipermeable membrane contains a solution in which the solute (that is dissolved in the liquid or solvent) is more in number than the solvent. At the outer sides of the semipermeable membrane, the only solvent is placed to witness the process of movement of solvent where it is more in number to the solution, where the solvent is less in number.
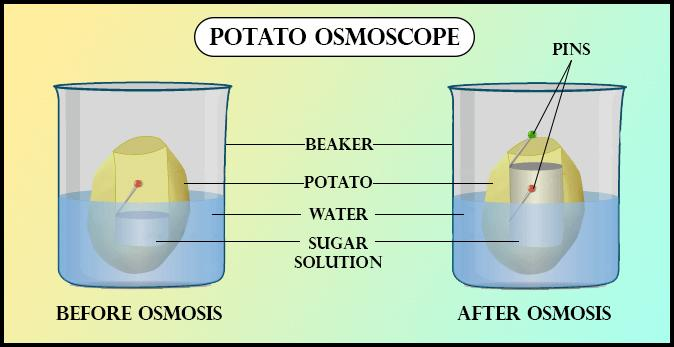
Example of potato osmoscope: Peel off the skin of a large-sized potato with the help of a scalpel. Cut its one end to make its base flat. Make a hollow cavity in the potato almost up to the bottom, Put the sugar solution into the cavity and mark the level by inserting a pin in the wall of the cavity of the potato. Place the potato in a beaker containing water. After sometime, it will be noticed that the level in the cavity rises. It is due to the phenomenon of osmosis. The experiments demonstrate that living cells of potato act as a differentially permeable membrane.
So, the correct answer is, ‘Demonstrating osmosis.’
Note: The other definition of osmosis can be the migration of solvent from a hypotonic solution (having a lower number of molecules of solute) to the hypertonic solution (having a higher number of molecules of solute) through a semipermeable membrane to keep the number of molecules on both sides equal.
Complete answer:
Osmoscope is an instrument used for the measurement of osmosis. The instrument contains a beaker into which a semipermeable membrane. The inner of the semipermeable membrane contains a solution in which the solute (that is dissolved in the liquid or solvent) is more in number than the solvent. At the outer sides of the semipermeable membrane, the only solvent is placed to witness the process of movement of solvent where it is more in number to the solution, where the solvent is less in number.
Example of potato osmoscope: Peel off the skin of a large-sized potato with the help of a scalpel. Cut its one end to make its base flat. Make a hollow cavity in the potato almost up to the bottom, Put the sugar solution into the cavity and mark the level by inserting a pin in the wall of the cavity of the potato. Place the potato in a beaker containing water. After sometime, it will be noticed that the level in the cavity rises. It is due to the phenomenon of osmosis. The experiments demonstrate that living cells of potato act as a differentially permeable membrane.
So, the correct answer is, ‘Demonstrating osmosis.’

Note: The other definition of osmosis can be the migration of solvent from a hypotonic solution (having a lower number of molecules of solute) to the hypertonic solution (having a higher number of molecules of solute) through a semipermeable membrane to keep the number of molecules on both sides equal.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Class 11 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Accountancy: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

In what year Guru Nanak Dev ji was born A15 April 1469 class 11 social science CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE




