Orlon has monomeric unit of :
(A) acrolein
(B) glycol
(C) vinyl cyanide
(D) isoprene
Answer
616.8k+ views
Hint: Orlon is the commercial name for the polymer polyacrylonitrile. Since orlon is a homo polymer, it is easy to find the repeating unit. The repeating unit found is the monomer for orlon also called as polyacrylonitrile.
Complete answer:
-Orlon is a synthetic acrylic fiber, which is used in the textile industry. The fiber is resistant to sunlight and atmospheric gases.
-This property of Orlon makes it suitable for outdoor uses. It is resistant to shrinkage and has a soft and warm feel. It is used in the manufacture of overcoating. Dress fabrics. Knitted wear, and washable woven sportswear.
-Orlon is an addition polymer. Let us draw the structure of Orlon to find the repeating unit.
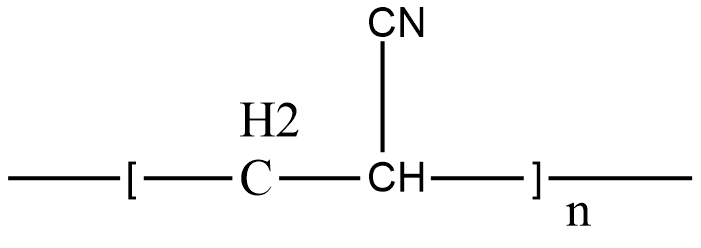
From the above structure we find that the repeating structure is acrylonitrile.
The common name for acrylonitrile is vinyl cyanide. This is because the common name for the nitrile functional group is cyanide and the nitrile functional group is attached at the vinyl position of the C=C double bond, hence the name vinyl cyanide.
The structure for vinyl cyanide is given below:
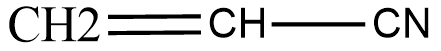
So, the correct answer is “Option C”.
Note: In the options given, acrolein is one of the options. Do not get confused between acrolein and acrylonitrile as acrolein has an aldehyde functional group and acryl nitrile has a nitrile functional group.
Complete answer:
-Orlon is a synthetic acrylic fiber, which is used in the textile industry. The fiber is resistant to sunlight and atmospheric gases.
-This property of Orlon makes it suitable for outdoor uses. It is resistant to shrinkage and has a soft and warm feel. It is used in the manufacture of overcoating. Dress fabrics. Knitted wear, and washable woven sportswear.
-Orlon is an addition polymer. Let us draw the structure of Orlon to find the repeating unit.

From the above structure we find that the repeating structure is acrylonitrile.
The common name for acrylonitrile is vinyl cyanide. This is because the common name for the nitrile functional group is cyanide and the nitrile functional group is attached at the vinyl position of the C=C double bond, hence the name vinyl cyanide.
The structure for vinyl cyanide is given below:
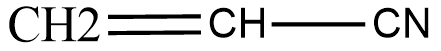
So, the correct answer is “Option C”.
Note: In the options given, acrolein is one of the options. Do not get confused between acrolein and acrylonitrile as acrolein has an aldehyde functional group and acryl nitrile has a nitrile functional group.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




