On reaction of chlorobenzene with acetyl chloride in presence of anhydrous \[AlC{l_3}\] the major product formed is.
Answer
551.4k+ views
Hint: We know that A Friedel-Crafts response is a natural coupling response including an electrophilic fragrant replacement that is utilized for the connection of substituents to sweet-smelling rings. The two essential sorts of Friedel-Crafts responses are the alkylation and acylation responses. These responses were created in the year \[1877\] by the French scientific expert Charles Friedel and the American physicist James Crafts.
Complete answer:
The Friedel-Crafts acylation response includes the expansion of an acyl gathering to a fragrant ring. Ordinarily, this is finished by utilizing a corrosive chloride \[\left( {R - \left( {C = O} \right) - {\text{ }}Cl} \right)\] and a Lewis corrosive impetus like\[AlC{l_3}\] . In a Friedel-Crafts acylation response, the sweet-smelling ring is changed into a ketone.
An acid anhydride can be utilized as an option in contrast to the acyl halide in Friedel-Crafts acylations. The halogen having a place with the acyl halide shapes a complex with the Lewis corrosive, producing a profoundly electrophilic acylium particle, which has an overall recipe of \[RC{O^ + }\] and is settled by reverberation.
The given reaction is Friedel Crafts acylation reaction and here para item structure as significant item due to ortho, para coordinating nature of \[C{l^ - }\] group. The major product formed by the given reaction is 2-chloroacetophenone.
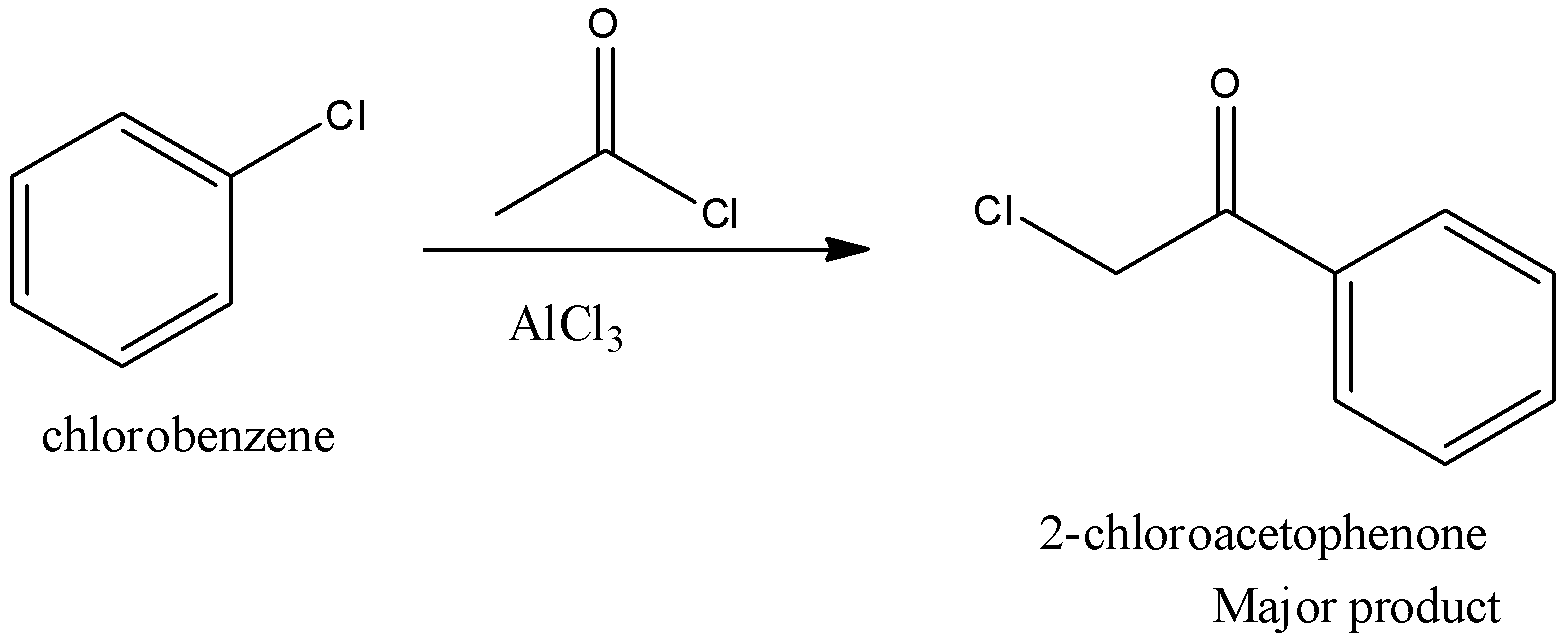
This reaction is drawn on chem.
Note:
We need to remember that the Friedel-Crafts Alkylation alludes to the supplanting of a fragrant proton with an alkyl bunch. This is done through an electrophilic assault on the sweet-smelling ring with the assistance of a carbocation. The Friedel-Crafts alkylation response is a technique for producing alkyl benzenes by utilizing alkyl halides as reactants. \[FeC{l_3}\] Or \[AlC{l_3}\] is utilized in this response to shape a carbocation by working with the evacuation of the halide. The subsequent carbocation goes through an improvement prior to continuing with the alkylation response.
Complete answer:
The Friedel-Crafts acylation response includes the expansion of an acyl gathering to a fragrant ring. Ordinarily, this is finished by utilizing a corrosive chloride \[\left( {R - \left( {C = O} \right) - {\text{ }}Cl} \right)\] and a Lewis corrosive impetus like\[AlC{l_3}\] . In a Friedel-Crafts acylation response, the sweet-smelling ring is changed into a ketone.
An acid anhydride can be utilized as an option in contrast to the acyl halide in Friedel-Crafts acylations. The halogen having a place with the acyl halide shapes a complex with the Lewis corrosive, producing a profoundly electrophilic acylium particle, which has an overall recipe of \[RC{O^ + }\] and is settled by reverberation.
The given reaction is Friedel Crafts acylation reaction and here para item structure as significant item due to ortho, para coordinating nature of \[C{l^ - }\] group. The major product formed by the given reaction is 2-chloroacetophenone.

This reaction is drawn on chem.
Note:
We need to remember that the Friedel-Crafts Alkylation alludes to the supplanting of a fragrant proton with an alkyl bunch. This is done through an electrophilic assault on the sweet-smelling ring with the assistance of a carbocation. The Friedel-Crafts alkylation response is a technique for producing alkyl benzenes by utilizing alkyl halides as reactants. \[FeC{l_3}\] Or \[AlC{l_3}\] is utilized in this response to shape a carbocation by working with the evacuation of the halide. The subsequent carbocation goes through an improvement prior to continuing with the alkylation response.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




