Number of $\sigma $ and $\pi $ bonds in ${{C}_{2}}$ molecule is/are?
A. 1$\sigma $ and 1$\pi $
B. 1$\sigma $ and 2 $\pi $
C. 2$\pi $
D. 1$\sigma $ and 3 $\pi $
Answer
606.9k+ views
Hint: Valence bond theory predicts covalent bond formation between atoms when they have half-filled valence atomic orbital, each containing a single unpaired electron. For example, sigma and pi bonds may overlap. Sigma bonds form when the two shared electrons have orbits that overlap head-to-head.
Complete Solution :
Diatomic carbon (${{C}_{2}}$) is a green, gaseous inorganic chemical. It is kinetically unstable at ambient temperature and pressure, being removed through auto polymerisation. Diatomic carbon is the second simplest form of carbon after atomic carbon. It is a gas that only exists above $3,642{}^\circ C$ below which it aggregates into graphite. It occurs in carbon vapour for example in blue hydrocarbon flames.
Now let us discuss the structure of ${{C}_{2}}$-
Molecular Orbital Theory shows that there are two sets of paired electrons in a degenerate $\pi $ bonding set of orbitals. Which means the bond order is 2. So there should exist a double bond between two carbon atoms in ${{C}_{2}}$ molecule. It is estimated that carbon vapour is around 28% diatomic, but theoretically it is dependent on the temperature and pressure. Their double bonds are made of two $\pi $ bonds because four electrons need to be accommodated in each bond. In bond formation only valence electrons or outermost electrons participate.
We can show the molecular orbital diagram pictorially as-
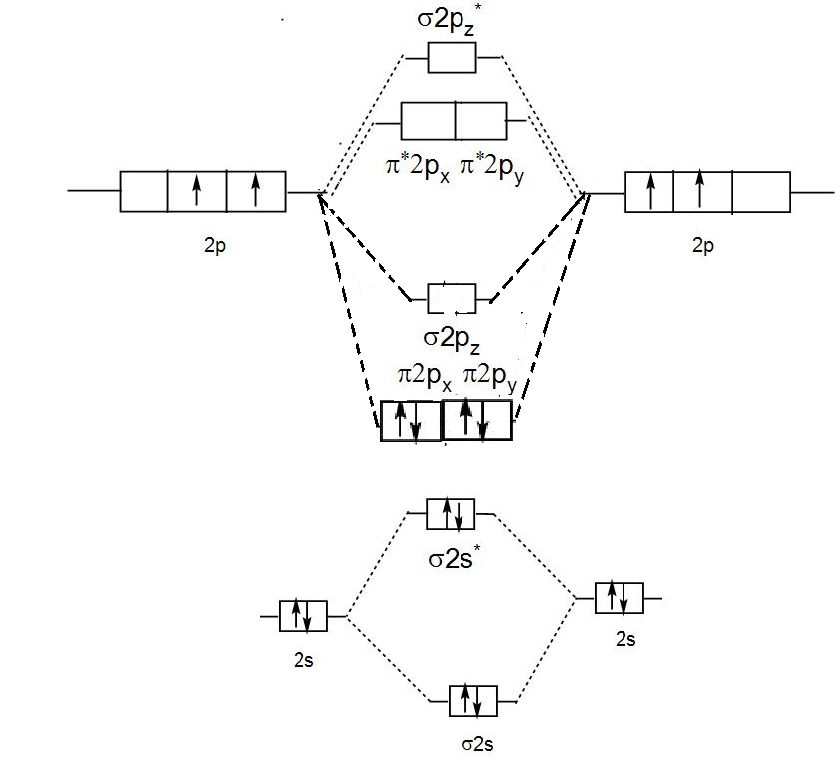
Hence, in ${{C}_{2}}$ molecules only 2$\pi $ are present.
So, the correct answer is “Option C”.
Note: We should know that sigma and pi are types of covalent bond.
Generally, there are three types of covalent bonding that we all know about. They are-
- Single bond- When there's a sharing of two electrons between a pair of atoms, it gives rise to a sigma bond between the atoms. The sigma bond thus formed is the single bond.
- Double bond- When four electrons are shared by the 2 atoms, it gives rise to a sigma bond and a pi-bond which we know as a double bond.
- Triple bond- When six electrons are shared by the two atoms, there exist one sigma and two pi-bonds thus forming a triple bond.
Complete Solution :
Diatomic carbon (${{C}_{2}}$) is a green, gaseous inorganic chemical. It is kinetically unstable at ambient temperature and pressure, being removed through auto polymerisation. Diatomic carbon is the second simplest form of carbon after atomic carbon. It is a gas that only exists above $3,642{}^\circ C$ below which it aggregates into graphite. It occurs in carbon vapour for example in blue hydrocarbon flames.
Now let us discuss the structure of ${{C}_{2}}$-
Molecular Orbital Theory shows that there are two sets of paired electrons in a degenerate $\pi $ bonding set of orbitals. Which means the bond order is 2. So there should exist a double bond between two carbon atoms in ${{C}_{2}}$ molecule. It is estimated that carbon vapour is around 28% diatomic, but theoretically it is dependent on the temperature and pressure. Their double bonds are made of two $\pi $ bonds because four electrons need to be accommodated in each bond. In bond formation only valence electrons or outermost electrons participate.
We can show the molecular orbital diagram pictorially as-

Hence, in ${{C}_{2}}$ molecules only 2$\pi $ are present.
So, the correct answer is “Option C”.
Note: We should know that sigma and pi are types of covalent bond.
Generally, there are three types of covalent bonding that we all know about. They are-
- Single bond- When there's a sharing of two electrons between a pair of atoms, it gives rise to a sigma bond between the atoms. The sigma bond thus formed is the single bond.
- Double bond- When four electrons are shared by the 2 atoms, it gives rise to a sigma bond and a pi-bond which we know as a double bond.
- Triple bond- When six electrons are shared by the two atoms, there exist one sigma and two pi-bonds thus forming a triple bond.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




