Number of equatorial bond in \[PC{l_5}\]is
Answer
620.1k+ views
Hint: We must know that the axial bond is longer than the equatorial bond because of greater repulsion from the equatorial bonds.
Complete step by step solution:
We must know that the bonds in an element to non-ring atoms are termed as axial or equatorial depending on their bond angle. The bonds to non-ring atoms with the angles of about 90° to that ring plane are termed as axial, whereas bonds to non-ring atoms which make only the small angle which is compared with the plane of the ring is termed as equatorial.
In Phosphorus pentachloride (\[PC{l_5}\]), there are 2 (two) axial bonds and 3 (three) equatorial bonds. The 2-axial bonds are at $90^o$ angle to the 3-equatorial bonds while all other equatorial bonds are at $120^o$ angle to each other atoms. So this axial bond is nearer to the equatorial bond and this causes a greater repulsion which then results in elongation of the bond length.
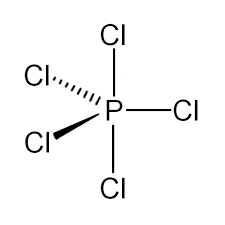
Therefore, we can conclude that the number of equatorial bonds in \[PC{l_5}\] is three.
Additional information: we must understand that the \[PC{l_5}\] has the trigonal bipyramidal structure. The hybridization is \[s{p_3}d\]which are hybrid orbitals occupied singly with Bond angles of $90^o$ and $120^o$.
Note: We must also know that in\[PC{l_5}\], the axial bond suffers repulsion from the equatorial bond so that they become weak and long but in \[P{F_5}\]the equatorial and axial bonds are of approx. nearly equal in length because the repulsion between these bonds tries to make axial bonds longer but fluorine being the most electronegative makes the above P-F bond shorter as a result of which axial bonds become nearly equal to the equatorial bonds.
Complete step by step solution:
We must know that the bonds in an element to non-ring atoms are termed as axial or equatorial depending on their bond angle. The bonds to non-ring atoms with the angles of about 90° to that ring plane are termed as axial, whereas bonds to non-ring atoms which make only the small angle which is compared with the plane of the ring is termed as equatorial.
In Phosphorus pentachloride (\[PC{l_5}\]), there are 2 (two) axial bonds and 3 (three) equatorial bonds. The 2-axial bonds are at $90^o$ angle to the 3-equatorial bonds while all other equatorial bonds are at $120^o$ angle to each other atoms. So this axial bond is nearer to the equatorial bond and this causes a greater repulsion which then results in elongation of the bond length.

Therefore, we can conclude that the number of equatorial bonds in \[PC{l_5}\] is three.
Additional information: we must understand that the \[PC{l_5}\] has the trigonal bipyramidal structure. The hybridization is \[s{p_3}d\]which are hybrid orbitals occupied singly with Bond angles of $90^o$ and $120^o$.
Note: We must also know that in\[PC{l_5}\], the axial bond suffers repulsion from the equatorial bond so that they become weak and long but in \[P{F_5}\]the equatorial and axial bonds are of approx. nearly equal in length because the repulsion between these bonds tries to make axial bonds longer but fluorine being the most electronegative makes the above P-F bond shorter as a result of which axial bonds become nearly equal to the equatorial bonds.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




