Number of double bond present in the Lewis structure of $ PF_6^ - $ is :
Answer
541.5k+ views
Hint: For hexafluorophosphate, [ $ P{F_6} $ ]-, there are six bonded groups and so no lone pairs. This anion is useful in synthesis as it also aids the crystallisation of bulky cations by having a suitable size match for the cation. The ion's negative charge is distributed through all seven atoms.
Complete answer:
The valence shell electron pair repulsion (VSEPR) theory is a mathematical model for predicting 3-D molecular geometry based on the number of valence shell electron bond pairs between atoms in a molecule or ion. As long as the central atom is not a metal, it can predict the form of almost all compounds with a central atom.
In this compound the central atom is phosphorus. We know that initially the valence electron on the central atom is $ 5 $ . The $ 6 $ Fluoride atom contributes one electron each. Hence, we add one negative charge to P. So the total valence electron is $ 12 $ .
Dividing $ 12 $ by $ 2 $ , we get $ 6 $ . Hence, the geometry formed in $ 6 $ electron pairs is Octahedral geometry. Therefore, The VSEPR model for $ {\left[ {P{F_6}} \right]^ - } $ is Octahedral.
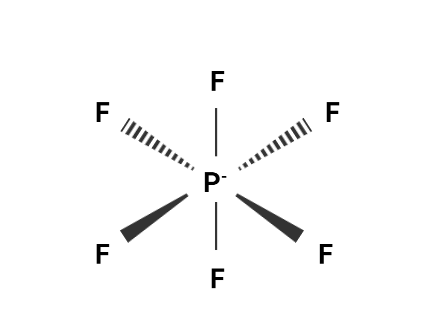
In this figure, we can conclude that there are no double bonds in the compound. Hence, the No. of double bonds present in the Lewis structure of $ PF_6^ - $ is $ 0 $ .
Note:
Hexafluorophosphate is most commonly used as the lithium salt, lithium hexafluorophosphate. This salt is a natural electrolyte in industrial secondary batteries, such as lithium-ion cells, when combined with dimethyl carbonate.
Complete answer:
The valence shell electron pair repulsion (VSEPR) theory is a mathematical model for predicting 3-D molecular geometry based on the number of valence shell electron bond pairs between atoms in a molecule or ion. As long as the central atom is not a metal, it can predict the form of almost all compounds with a central atom.
In this compound the central atom is phosphorus. We know that initially the valence electron on the central atom is $ 5 $ . The $ 6 $ Fluoride atom contributes one electron each. Hence, we add one negative charge to P. So the total valence electron is $ 12 $ .
Dividing $ 12 $ by $ 2 $ , we get $ 6 $ . Hence, the geometry formed in $ 6 $ electron pairs is Octahedral geometry. Therefore, The VSEPR model for $ {\left[ {P{F_6}} \right]^ - } $ is Octahedral.

In this figure, we can conclude that there are no double bonds in the compound. Hence, the No. of double bonds present in the Lewis structure of $ PF_6^ - $ is $ 0 $ .
Note:
Hexafluorophosphate is most commonly used as the lithium salt, lithium hexafluorophosphate. This salt is a natural electrolyte in industrial secondary batteries, such as lithium-ion cells, when combined with dimethyl carbonate.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




