Nitrosyl ligand binds to d-metal atoms in linear and bent fashion and behaves , respectively as
A.$N{O^ + }$ and $N{O^ + }$
B.$N{O^ + }$ and $N{O^ - }$
C.$N{O^ - }$ and $N{O^ - }$
D.$N{O^ - }$ and $N{O^ + }$
Answer
583.5k+ views
Hint:Nitrosyl complexes or metal nitrosyl complexes are complexes which contain nitric oxide, $NO$ bonded to the transitional metal. Of the many kinds of the nitrosyl complexes, they differ both in co ligand and structure.
Complete step by step answer:
Most complexes which have $NO$ ligand may be seen or viewed as a nitrosyl cation $N{O^ + }$ . It feeds two electrons to the metal and in return accepts electrons from the metal via back-bonding.
The \[M - N - O\] present in nitrosyl complexes are usually linear, or have no more than \[15^\circ \] from linear. But in some complexes, however, it is noticed, especially when back-bonding is a little less important, the \[M - N - O\] angle can strongly deviate from \[180^\circ \] .
We will see that, in \[N{O^ + }\] structure, there exists a triple bond between $N$ and ${O^ - }$ atoms and two electrons which act as a lone pair of electrons or coordinate, so the angle between \[M - N - O\] is \[180^\circ \]
While in \[N{O^ - }\] structure double bond exist between $N$ and ${O^ - }$ atoms while the \[4{\text{ }}{e^\_}\] acts as two lone pairs of electrons , so only one lone pair of electron coordinate with metal atom.
Suppose for a metal $M$ the bonding with $N{O^ + }$ goes as -
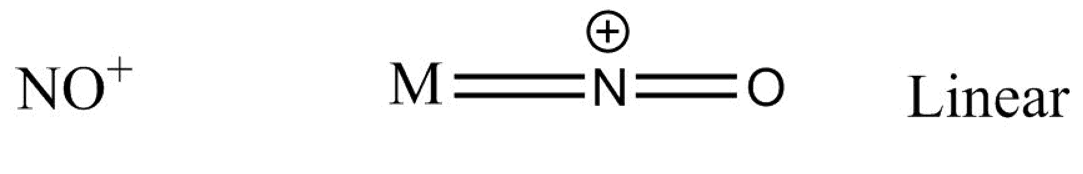
Which gives us a linear structure.
And bonding with $N{O^ - }$ goes as –
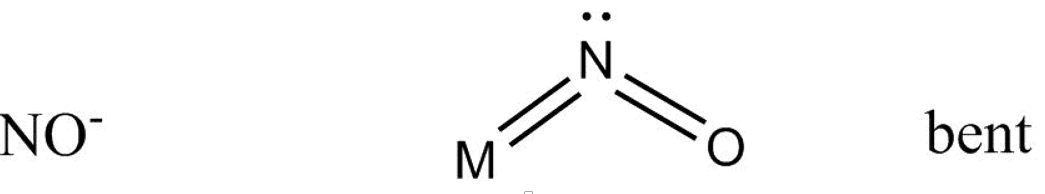
Which gives us a bent structure.
Therefore we can conclude that for the mentioned question,
Option (B). $N{O^ + }$ and $N{O^ - }$ is the correct answer.
Additional information –
The linear and bent \[NO\] ligands can be distinguished using infrared spectroscopy. Linear \[M - N - O\] groups absorb in the range \[1650-1900{\text{ }}c{m^{ - 1}}\], where as, the bent nitrosyls tend to absorb in the range of \[1525-1690{\text{ }}c{m^{ - 1}}\]. They differ in their vibrational frequencies and it reflects the differing \[N - O\] bond orders for linear (triple bond) and bent \[NO\] (double bond).
Note:
The bent $NO$ ligand is sometimes described as the anion, \[N{O^ - }\]. Examples for such compounds are the organic nitroso compounds, such as nitrosobenzene. For example a complex with a bent \[NO\] ligand is trans\[ - {\left[ {Co{{\left( {en} \right)}_2}\left( {NO} \right)Cl} \right]^ + }\]
Complete step by step answer:
Most complexes which have $NO$ ligand may be seen or viewed as a nitrosyl cation $N{O^ + }$ . It feeds two electrons to the metal and in return accepts electrons from the metal via back-bonding.
The \[M - N - O\] present in nitrosyl complexes are usually linear, or have no more than \[15^\circ \] from linear. But in some complexes, however, it is noticed, especially when back-bonding is a little less important, the \[M - N - O\] angle can strongly deviate from \[180^\circ \] .
We will see that, in \[N{O^ + }\] structure, there exists a triple bond between $N$ and ${O^ - }$ atoms and two electrons which act as a lone pair of electrons or coordinate, so the angle between \[M - N - O\] is \[180^\circ \]
While in \[N{O^ - }\] structure double bond exist between $N$ and ${O^ - }$ atoms while the \[4{\text{ }}{e^\_}\] acts as two lone pairs of electrons , so only one lone pair of electron coordinate with metal atom.
Suppose for a metal $M$ the bonding with $N{O^ + }$ goes as -

Which gives us a linear structure.
And bonding with $N{O^ - }$ goes as –

Which gives us a bent structure.
Therefore we can conclude that for the mentioned question,
Option (B). $N{O^ + }$ and $N{O^ - }$ is the correct answer.
Additional information –
The linear and bent \[NO\] ligands can be distinguished using infrared spectroscopy. Linear \[M - N - O\] groups absorb in the range \[1650-1900{\text{ }}c{m^{ - 1}}\], where as, the bent nitrosyls tend to absorb in the range of \[1525-1690{\text{ }}c{m^{ - 1}}\]. They differ in their vibrational frequencies and it reflects the differing \[N - O\] bond orders for linear (triple bond) and bent \[NO\] (double bond).
Note:
The bent $NO$ ligand is sometimes described as the anion, \[N{O^ - }\]. Examples for such compounds are the organic nitroso compounds, such as nitrosobenzene. For example a complex with a bent \[NO\] ligand is trans\[ - {\left[ {Co{{\left( {en} \right)}_2}\left( {NO} \right)Cl} \right]^ + }\]
Recently Updated Pages
Master Class 10 Computer Science: Engaging Questions & Answers for Success

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Master Class 10 Science: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




