Melting point of phenol is:
$A.\;{95^ \circ }C$
$B.\;{43^ \circ }C$
$C.\;{20^ \circ }C$
$D.\;{10^ \circ }C$
Answer
619.5k+ views
Hint: An organic compound-Phenol is appreciably soluble in water and because of its commercial importance many methods have been developed for its production. They are generally colourless solids or liquids and have relatively low melting point.
Complete answer:
Phenol is also known as carbolic acid or Hydroxybenzene or Phenic acid. And its molecular formula is ${C_6}{H_5}OH$. It is an aromatic organic compound. It is a white crystalline solid that is volatile. The molecule of Phenol consists of a phenyl group $( - {C_6}{H_5})$which is bonded to a hydroxyl group $( - OH)$. It requires careful handling because it is mildly acidic and can cause chemical burns. In aqueous solution the pH range is 8 – 12 pH. And its melting point is ${43^ \circ }C$
Phenol was first extracted from coal tar, but today it is produced on a large scale which is around 7 billion kg/year from petroleum derived feedstocks.
Also, it is used to synthesize plastics and related materials.
The structure of Phenol is
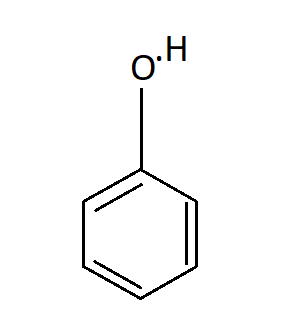
Hence our answer is B. ${43^ \circ }C$
Note: Phenol is a colourless to white solid when pure. It has a distinct odour which is sickeningly sweet and tarry. We can taste and smell phenol at levels lower than those that are associated with harmful effects. It evaporates more slowly than water, and a moderate amount can form a solution with water. Also, they can catch fire.
Complete answer:
Phenol is also known as carbolic acid or Hydroxybenzene or Phenic acid. And its molecular formula is ${C_6}{H_5}OH$. It is an aromatic organic compound. It is a white crystalline solid that is volatile. The molecule of Phenol consists of a phenyl group $( - {C_6}{H_5})$which is bonded to a hydroxyl group $( - OH)$. It requires careful handling because it is mildly acidic and can cause chemical burns. In aqueous solution the pH range is 8 – 12 pH. And its melting point is ${43^ \circ }C$
Phenol was first extracted from coal tar, but today it is produced on a large scale which is around 7 billion kg/year from petroleum derived feedstocks.
Also, it is used to synthesize plastics and related materials.
The structure of Phenol is

Hence our answer is B. ${43^ \circ }C$
Note: Phenol is a colourless to white solid when pure. It has a distinct odour which is sickeningly sweet and tarry. We can taste and smell phenol at levels lower than those that are associated with harmful effects. It evaporates more slowly than water, and a moderate amount can form a solution with water. Also, they can catch fire.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which gas is abundant in air class 11 chemistry CBSE

10 examples of friction in our daily life




