What is meant by the chelate effect? Give an example.
Answer
617.1k+ views
Hint: Chelate is a term related to coordination compounds. When a di- or polydentate ligand uses its two or more donor atoms together to bind a single metal ion then it is said to be a chelate ligand. The number
of such ligating groups is called denticity of the ligand.
Complete answer: When a ligand is bound to a metal ion through one single donor atom then it is called unidetate but when it uses two donor atoms it is said to be a bidentate ligand. $C{l^ - }$ , ${H_2}O$are unidentate ligands and ${C_2}O_4^ - $ , ${H_2}NC{H_2}C{H_2}N{H_2}$are bidentate ligands. Similarly ligands having more than two donor atoms are called polydentate ligands $N{\left( {C{H_2}C{H_2}N{H_2}} \right)_3}$is a polydentate ligand. When a di- or polydentate ligand uses its two or more donor atoms together to bind a single metal ion then it is said to be a chelate ligand. When a ligand uses its two or more donor atoms simultaneously a ring-like structure is formed. The formation of such ring-like structures is called a chelate effect. Compounds having chelate rings are more stable as compared to other compounds. An example of such a complex is ethylenediamine-cadmium. The structure of this complex is:
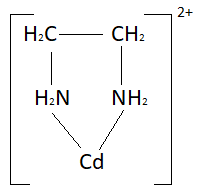
Note:
The coordination number of the metal complex is defined as the number of ligand donor atoms to which metal is directly bonded. The central atom/ion and the ligands attached to it are enclosed in a square bracket and the term collectively used is the coordination sphere.
of such ligating groups is called denticity of the ligand.
Complete answer: When a ligand is bound to a metal ion through one single donor atom then it is called unidetate but when it uses two donor atoms it is said to be a bidentate ligand. $C{l^ - }$ , ${H_2}O$are unidentate ligands and ${C_2}O_4^ - $ , ${H_2}NC{H_2}C{H_2}N{H_2}$are bidentate ligands. Similarly ligands having more than two donor atoms are called polydentate ligands $N{\left( {C{H_2}C{H_2}N{H_2}} \right)_3}$is a polydentate ligand. When a di- or polydentate ligand uses its two or more donor atoms together to bind a single metal ion then it is said to be a chelate ligand. When a ligand uses its two or more donor atoms simultaneously a ring-like structure is formed. The formation of such ring-like structures is called a chelate effect. Compounds having chelate rings are more stable as compared to other compounds. An example of such a complex is ethylenediamine-cadmium. The structure of this complex is:

Note:
The coordination number of the metal complex is defined as the number of ligand donor atoms to which metal is directly bonded. The central atom/ion and the ligands attached to it are enclosed in a square bracket and the term collectively used is the coordination sphere.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Class 11 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Accountancy: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

In what year Guru Nanak Dev ji was born A15 April 1469 class 11 social science CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE




