In the compound between A and H, what type of bond is formed?
(A) Ionic
(B) Covalent
(C) Coordinate
(D) No bond is formed
Answer
605.1k+ views
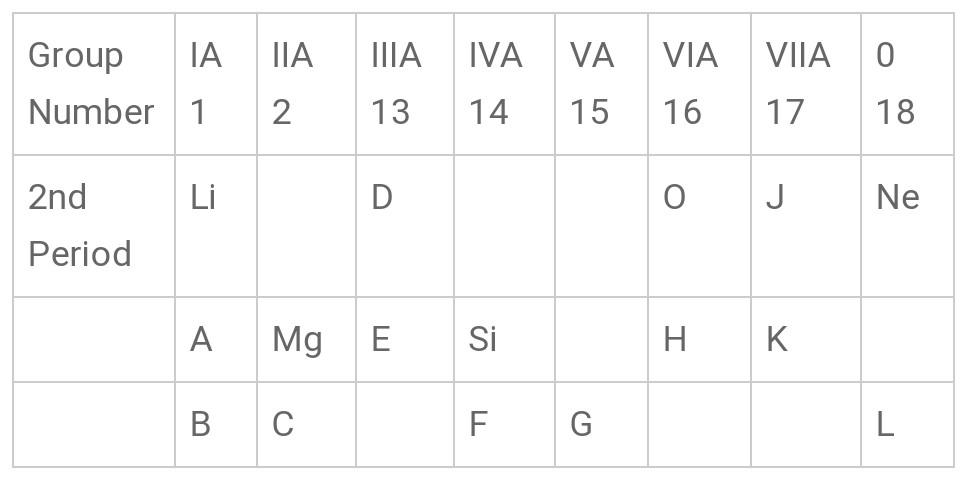
Hint: By looking in the below table, we would get an idea about which element is A and which is H. Determining their nature we can easily say what type of bond is formed between them.
Complete step by step solution:
From the table given above, we can see that A belongs to Group IA and H belongs to Group VIA. Since A is below Lithium (Li-Atomic No.3) and next Neon (Atomic No. 10), it must surely be Sodium (Na- Atomic No. 11). H is placed in Group VIA, before the potassium (K- Atomic No. 17), so it must be Sulphur (S) with Atomic Number 16. As we know Na is a metal and S is a Nonmetal, the bond formed between them must be an ionic bond and the compound is called Ionic compound.
Definition: Ionic bond is the force of attraction between a positive and negative ion. This bond will be formed when the metallic atom gives its electrons to the nonmetallic atom.
During this time metal will give its electrons to the nonmetal, in order to obtain the nearest stable electronic configuration. Similarly, nonmetal will also accept those electrons from the metals in order to obtain the nearest stable electronic configuration.
$2Na+S\to N{{a}_{2}}S$
Sodium metal by giving its electron will attain [Ne] configuration and Sulphur will obtain two electrons (one from each Na metal) to attain [Ar] configuration.
Therefore, the correct answer is option (A) Ionic.
Additional Information:
There are various other bonds. They are
- Covalent bond
- Coordinate bond
- Hydrogen bond
Note: Covalent bonds will be formed only between two nonmetals. Ionic bonds are formed between a metal and a nonmetal. Strength of ionic bonds depends on the difference of electronegativity of the bonded atoms.

Complete step by step solution:
From the table given above, we can see that A belongs to Group IA and H belongs to Group VIA. Since A is below Lithium (Li-Atomic No.3) and next Neon (Atomic No. 10), it must surely be Sodium (Na- Atomic No. 11). H is placed in Group VIA, before the potassium (K- Atomic No. 17), so it must be Sulphur (S) with Atomic Number 16. As we know Na is a metal and S is a Nonmetal, the bond formed between them must be an ionic bond and the compound is called Ionic compound.
Definition: Ionic bond is the force of attraction between a positive and negative ion. This bond will be formed when the metallic atom gives its electrons to the nonmetallic atom.
During this time metal will give its electrons to the nonmetal, in order to obtain the nearest stable electronic configuration. Similarly, nonmetal will also accept those electrons from the metals in order to obtain the nearest stable electronic configuration.
$2Na+S\to N{{a}_{2}}S$
Sodium metal by giving its electron will attain [Ne] configuration and Sulphur will obtain two electrons (one from each Na metal) to attain [Ar] configuration.
Therefore, the correct answer is option (A) Ionic.
Additional Information:
There are various other bonds. They are
- Covalent bond
- Coordinate bond
- Hydrogen bond
Note: Covalent bonds will be formed only between two nonmetals. Ionic bonds are formed between a metal and a nonmetal. Strength of ionic bonds depends on the difference of electronegativity of the bonded atoms.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




