In Rosenmund reactions, presence of \[BaS{O_4}\]acts as _______ for \[Pd\].
(A): promoter
(B): moderator
(C): inhibitor
(D): poison
Answer
584.7k+ views
Hint: A Rosenmund reaction can be referred to as a hydrogenation process which involves a reaction between molecular hydrogen and acyl chloride in the presence of palladium (catalyst) on barium sulphate (\[BaS{O_4}\]). The mechanism of this reaction mainly explains how acyl chlorides get selectively reduced to aldehydes. This reaction is named after Karl Wilhelm Rosenmund who reported this reaction for the first time in 1918.
Complete step by step answer:
A Rosenmund reaction or Rosenmund Reduction, an organic chemical reaction, converts an acid chloride into an aldehyde by passing the hydrogen gas over \[Pd\] (palladium) on \[BaS{O_4}\]. Actually, \[BaS{O_4}\] decreases the activity of \[Pd\] (or partially deactivate \[Pd\]) owing to its very low surface area. Thereby, it also declines the reducing power of \[Pd\]to prevent the over-reduction of acid. Hence, \[BaS{O_4}\] acts as a poison for \[Pd\]. If reducing power of \[Pd\ ] is not reduced, it can further reduce aldehyde to alcohols which can ultimately produce esters by reacting with remaining acyl chloride. The Rosenmund reaction can be written as stated below:
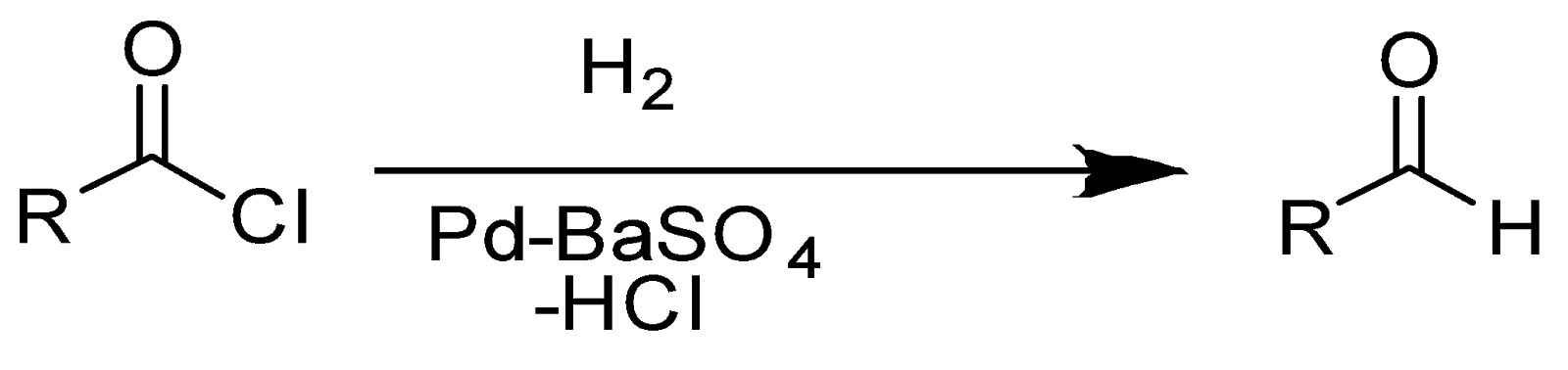
An example of Rosenmund reaction is: reaction of benzoyl chloride (which is an acid chloride) with molecular hydrogen as well as \[Pd\]/\[BaS{O_4}\](which is a poisoned catalyst) producing benzaldehyde.
Thus, the correct answer is Option D.
Note: For certain highly reactive acyl chlorides, the Rosenmund catalyst (\[Pd\]-\[BaS{O_4}\]) has to be mixed with more poisons such as thioquinanthrene or thiourea to prevent further reduction or hydrogenation. Apart from this, Formaldehyde cannot be synthesized through this method as formyl chloride being unstable at room temperature. Application of Rosenmund reaction is formation of saturated fatty aldehyde.
Complete step by step answer:
A Rosenmund reaction or Rosenmund Reduction, an organic chemical reaction, converts an acid chloride into an aldehyde by passing the hydrogen gas over \[Pd\] (palladium) on \[BaS{O_4}\]. Actually, \[BaS{O_4}\] decreases the activity of \[Pd\] (or partially deactivate \[Pd\]) owing to its very low surface area. Thereby, it also declines the reducing power of \[Pd\]to prevent the over-reduction of acid. Hence, \[BaS{O_4}\] acts as a poison for \[Pd\]. If reducing power of \[Pd\ ] is not reduced, it can further reduce aldehyde to alcohols which can ultimately produce esters by reacting with remaining acyl chloride. The Rosenmund reaction can be written as stated below:

An example of Rosenmund reaction is: reaction of benzoyl chloride (which is an acid chloride) with molecular hydrogen as well as \[Pd\]/\[BaS{O_4}\](which is a poisoned catalyst) producing benzaldehyde.
Thus, the correct answer is Option D.
Note: For certain highly reactive acyl chlorides, the Rosenmund catalyst (\[Pd\]-\[BaS{O_4}\]) has to be mixed with more poisons such as thioquinanthrene or thiourea to prevent further reduction or hydrogenation. Apart from this, Formaldehyde cannot be synthesized through this method as formyl chloride being unstable at room temperature. Application of Rosenmund reaction is formation of saturated fatty aldehyde.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




