In a conductometric titration experiment, a solution of $0.1MBa{\left( {OH} \right)_2}$ is titrated against a solution of $0.1MMg\left( {S{O_4}} \right)$ and the conductance of the mixture is continuously measured. The correct variation of conductance of the reaction mixture with the titration volume of $MgS{O_4}$ is best represented by.
A)
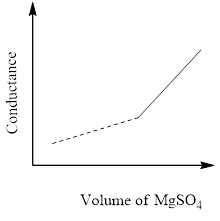
B)
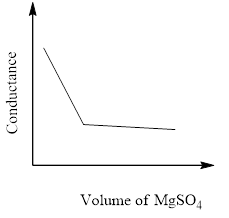
C)
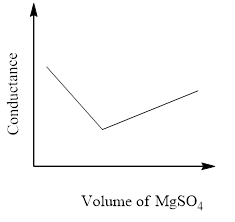
D)
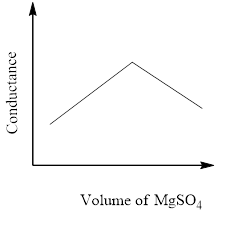 .
.




Answer
600k+ views
Hint: We know that the conductometric titration may be a laboratory method of quantitative chemical analysis wont to identify the concentration of a given analyte during a mixture. Conductometric titration involves the frequent adding of a reactant to a reaction mixture and consequently the records of the resultant change within the electrolytic conductivity of the reaction mixture
Complete step by step answer:
First we see the principle of the conductometric titration.
The principle of the conductometric titration is in a titration process, one ion is replaced with another and therefore the variation within the ionic conductivities of those ions directly impacts on the common electrolytic conductivity of the solution.
The conductivity that's measured in an electrolyte solution depends on the sort and concentration of the ions. As long since the reaction is taking its course the conductivity drops when the quality solution is in excess, the conductivity rises again.
So, the correct answer is Option C.
Note:
Now we can discuss about the advantages and drawbacks of conductometric titration as,
Advantages Conductometric Titration:
This process is extremely useful within the titrations of very dilute solutions and weak acids.
The end-point of this method of titration is extremely sharp and accurate in comparison to a couple of other titration processes.
This type of titration is applicable for solutions that are colored or turbid, and that the endpoint of the titration with normal indicators can't be observed easily by the human eye.
Conductometric titration has numerous applications in acid-base titrations, redox titrations, precipitation titrations, and sophisticated titrations.
The major drawbacks of this sort of titration include:
Only a couple of specific redox titrations are often through with the assistance of this process. This is often because the conductivity of the answer is masked by relatively high hydronium ion concentration.
Complete step by step answer:
First we see the principle of the conductometric titration.
The principle of the conductometric titration is in a titration process, one ion is replaced with another and therefore the variation within the ionic conductivities of those ions directly impacts on the common electrolytic conductivity of the solution.
The conductivity that's measured in an electrolyte solution depends on the sort and concentration of the ions. As long since the reaction is taking its course the conductivity drops when the quality solution is in excess, the conductivity rises again.
So, the correct answer is Option C.
Note:
Now we can discuss about the advantages and drawbacks of conductometric titration as,
Advantages Conductometric Titration:
This process is extremely useful within the titrations of very dilute solutions and weak acids.
The end-point of this method of titration is extremely sharp and accurate in comparison to a couple of other titration processes.
This type of titration is applicable for solutions that are colored or turbid, and that the endpoint of the titration with normal indicators can't be observed easily by the human eye.
Conductometric titration has numerous applications in acid-base titrations, redox titrations, precipitation titrations, and sophisticated titrations.
The major drawbacks of this sort of titration include:
Only a couple of specific redox titrations are often through with the assistance of this process. This is often because the conductivity of the answer is masked by relatively high hydronium ion concentration.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




