Give the structural formula and IUPAC names of the following compounds
(a) Malonic acid
(b) Succinic acid.
Answer
594k+ views
Hint:Malonic acid and succinic acid are dicarboxylic acids belonging to a homologous group, that is, compounds which differ only by a $ - C{H_2}$ group and molecular mass of $14$. Malonic acid is the second member of this series while succinic acid is the third member.
Complete step by step answer:
Both malonic acid and succinic acid belong to dicarboxylic acids, meaning that they have two carboxylic acid ($ - COOH$) groups in their structure. They are also members of a homologous series, which are compounds which differ only by a $ - C{H_2}$ group and molecular mass of $14$. The first member of this series is oxalic acid, which has only two $ - COOH$ groups. Malonic acid being the next member, has a $ - C{H_2}$ group in between the two carboxylic acid groups, while succinic acid, the next member, has two $ - C{H_2}$ groups. With this information, we can draw their structure as follows:
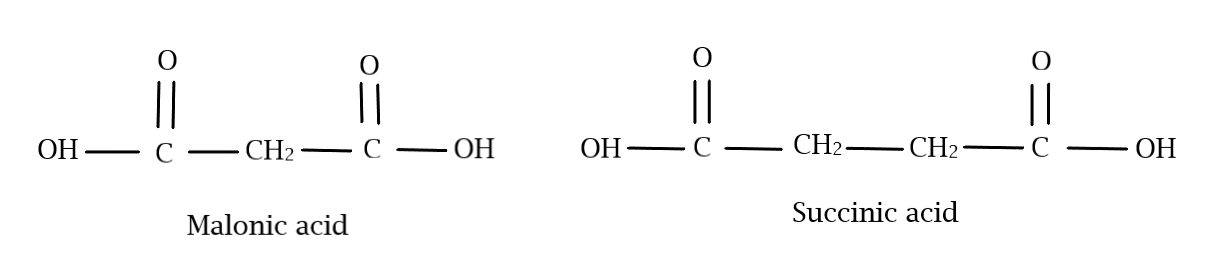
For the IUPAC name of malonic acid, let us start counting the carbon atoms from the left. We have 3 carbon atoms and two carboxylic acid groups on the first and third carbon atoms. For the three carbon atoms, we have to include “propane” and to account for the two carboxylic acids, we include “dioic acid” with the numbers at which they are present written at the beginning. Hence the IUPAC name is:
$1,3$ – propanedioic acid
For the IUPAC name of succinic acid, let us start counting the carbon atoms from the left. We have 4 carbon atoms and two carboxylic acid groups on the first and fourth carbon atoms. For the four carbon atoms, we have to include “butane” and to account for the two carboxylic acids, we include “dioic acid” with the numbers at which they are present written at the beginning. Hence the IUPAC name is:
$1,4$ – butanedioic acid
Note:
While writing IUPAC name, numbering of carbon atoms should begin from the closest carbon atom to the functional group. Here as both the compounds are symmetric (same groups of atoms on either side of a line drawn through the middle of the compound) and contain the same functional group, we can start numbering from any side and still get the same answer. When two or more functional groups are present, numbering should be done according to the series of preference of functional groups.
Complete step by step answer:
Both malonic acid and succinic acid belong to dicarboxylic acids, meaning that they have two carboxylic acid ($ - COOH$) groups in their structure. They are also members of a homologous series, which are compounds which differ only by a $ - C{H_2}$ group and molecular mass of $14$. The first member of this series is oxalic acid, which has only two $ - COOH$ groups. Malonic acid being the next member, has a $ - C{H_2}$ group in between the two carboxylic acid groups, while succinic acid, the next member, has two $ - C{H_2}$ groups. With this information, we can draw their structure as follows:
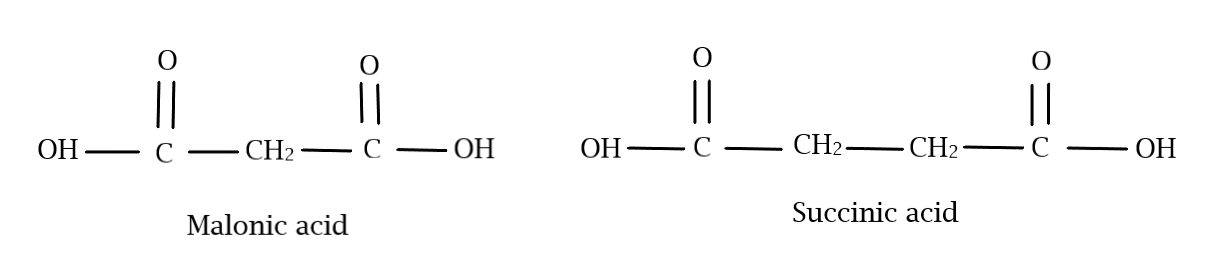
For the IUPAC name of malonic acid, let us start counting the carbon atoms from the left. We have 3 carbon atoms and two carboxylic acid groups on the first and third carbon atoms. For the three carbon atoms, we have to include “propane” and to account for the two carboxylic acids, we include “dioic acid” with the numbers at which they are present written at the beginning. Hence the IUPAC name is:
$1,3$ – propanedioic acid
For the IUPAC name of succinic acid, let us start counting the carbon atoms from the left. We have 4 carbon atoms and two carboxylic acid groups on the first and fourth carbon atoms. For the four carbon atoms, we have to include “butane” and to account for the two carboxylic acids, we include “dioic acid” with the numbers at which they are present written at the beginning. Hence the IUPAC name is:
$1,4$ – butanedioic acid
Note:
While writing IUPAC name, numbering of carbon atoms should begin from the closest carbon atom to the functional group. Here as both the compounds are symmetric (same groups of atoms on either side of a line drawn through the middle of the compound) and contain the same functional group, we can start numbering from any side and still get the same answer. When two or more functional groups are present, numbering should be done according to the series of preference of functional groups.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




