Gem dihalide on hydrolysis gives:
a.) vicinal diol
b.) geminal diol
c.) carbonyl compound
d.) carboxylic acid
Answer
627.3k+ views
Hint: Geminal dihalide hydrolysis is an organic reaction in which the gem-halide reacts with water or undergo hydrolysis in an alkaline medium. There are two products which can be formed.
Complete step by step solution: Geminal dihalides are dihalogen compounds in which both the halogen atoms are attached to the same carbon atom.
For example, Ethylidene dichloride
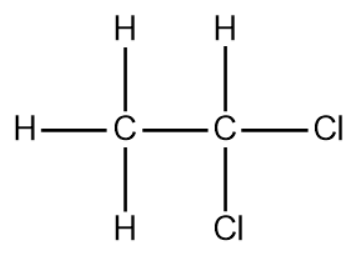
Suppose, there is a geminal dihalide \[C{{H}_{3}}CH{{(Cl)}_{2}}\] is present, the hydrolysis of gem-halides takes place in alkaline medium. So, we will hydrolyse it in the presence of aqueous NaOH/KOH.
On hydrolysis, both of the chlorine group will be replaced by hydroxyl group (OH),
\[C{{H}_{3}}CH{{(OH)}_{2}}\] will be formed.
As both the hydroxyl groups are present on the same carbon, this compound is not stable. So, water acts as a leaving group i.e. \[{{H}_{2}}O\] will be removed.
Thus, forming an aldehyde group \[C{{H}_{3}}CHO\], this compound is called acetaldehyde. But this is the case when gem-dihalide is present at the terminal position.
\[C{{H}_{3}}CH{{(Cl)}_{2}}\xrightarrow[aq]{KOH}C{{H}_{3}}CH{{(OH)}_{2}}\to C{{H}_{3}}COH+{{H}_{2}}O\]
Now, let's take a compound \[C{{H}_{3}}C{{(Cl)}_{2}}C{{H}_{3}}\], on hydrolysis the chlorine groups will be replaced by hydroxyl groups, \[C{{H}_{3}}C{{(OH)}_{2}}C{{H}_{3}}\]will be obtained.
Similarly, as the previous example, this compound will also be unstable, thus water will act as a leaving group.
Thus, forming a keto or carbonyl group \[C{{H}_{3}}COC{{H}_{3}}\], this compound is called acetone.
\[C{{H}_{3}}C{{(Cl)}_{2}}C{{H}_{3}}\xrightarrow[aq]{KOH}C{{H}_{3}}COC{{H}_{3}}+2KCl+{{H}_{2}}O\]
Therefore, from the above statements we can conclude that the correct option is (c).
Note: Geminal dihalides are prepared by reacting a non-enolizable aldehyde and/or ketone with phosgene or thionyl chloride in the presence of an organic-phosphorus compound.
Complete step by step solution: Geminal dihalides are dihalogen compounds in which both the halogen atoms are attached to the same carbon atom.
For example, Ethylidene dichloride

Suppose, there is a geminal dihalide \[C{{H}_{3}}CH{{(Cl)}_{2}}\] is present, the hydrolysis of gem-halides takes place in alkaline medium. So, we will hydrolyse it in the presence of aqueous NaOH/KOH.
On hydrolysis, both of the chlorine group will be replaced by hydroxyl group (OH),
\[C{{H}_{3}}CH{{(OH)}_{2}}\] will be formed.
As both the hydroxyl groups are present on the same carbon, this compound is not stable. So, water acts as a leaving group i.e. \[{{H}_{2}}O\] will be removed.
Thus, forming an aldehyde group \[C{{H}_{3}}CHO\], this compound is called acetaldehyde. But this is the case when gem-dihalide is present at the terminal position.
\[C{{H}_{3}}CH{{(Cl)}_{2}}\xrightarrow[aq]{KOH}C{{H}_{3}}CH{{(OH)}_{2}}\to C{{H}_{3}}COH+{{H}_{2}}O\]
Now, let's take a compound \[C{{H}_{3}}C{{(Cl)}_{2}}C{{H}_{3}}\], on hydrolysis the chlorine groups will be replaced by hydroxyl groups, \[C{{H}_{3}}C{{(OH)}_{2}}C{{H}_{3}}\]will be obtained.
Similarly, as the previous example, this compound will also be unstable, thus water will act as a leaving group.
Thus, forming a keto or carbonyl group \[C{{H}_{3}}COC{{H}_{3}}\], this compound is called acetone.
\[C{{H}_{3}}C{{(Cl)}_{2}}C{{H}_{3}}\xrightarrow[aq]{KOH}C{{H}_{3}}COC{{H}_{3}}+2KCl+{{H}_{2}}O\]
Therefore, from the above statements we can conclude that the correct option is (c).
Note: Geminal dihalides are prepared by reacting a non-enolizable aldehyde and/or ketone with phosgene or thionyl chloride in the presence of an organic-phosphorus compound.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




