Formula for ferrocene is
A.\[{\left[ {{\text{Fe(CN}}{{\text{)}}_{\text{6}}}} \right]^{{\text{4 - }}}}\]
B.\[{\left[ {{\text{Fe(CN}}{{\text{)}}_{\text{6}}}} \right]^{{\text{3 + }}}}\]
C.\[\left[ {{\text{Fe(CO}}{{\text{)}}_{\text{5}}}} \right]\]
D.\[\left[ {{\text{Fe(}}{{\text{C}}_{\text{5}}}{{\text{H}}_{\text{5}}}{{\text{)}}_{\text{2}}}} \right]\]
Answer
585k+ views
Hint:Ferrocene is actually a metallocene or we can say the first sandwich compound. Ferrocenes are the molecules where metal atoms are sandwiched between two parallel and planar cyclopentadienyl rings. Organometallic compound of \[{\text{Fe}}\] is ferrocene.
Complete step by step answer:
In ferrocene \[{\text{Fe}}\] metal is attached with a ligand called cyclopentadiene, the formula for cyclopentadiene is \[{{\text{C}}_{\text{5}}}{{\text{H}}_{\text{5}}}\]. In ferrocene, two cyclopentadienyl ligand present so the formula is \[\left[ {{\text{Fe(}}{{\text{C}}_{\text{5}}}{{\text{H}}_{\text{5}}}{{\text{)}}_{\text{2}}}} \right]\]. Two important conformers of ferrocene are: -
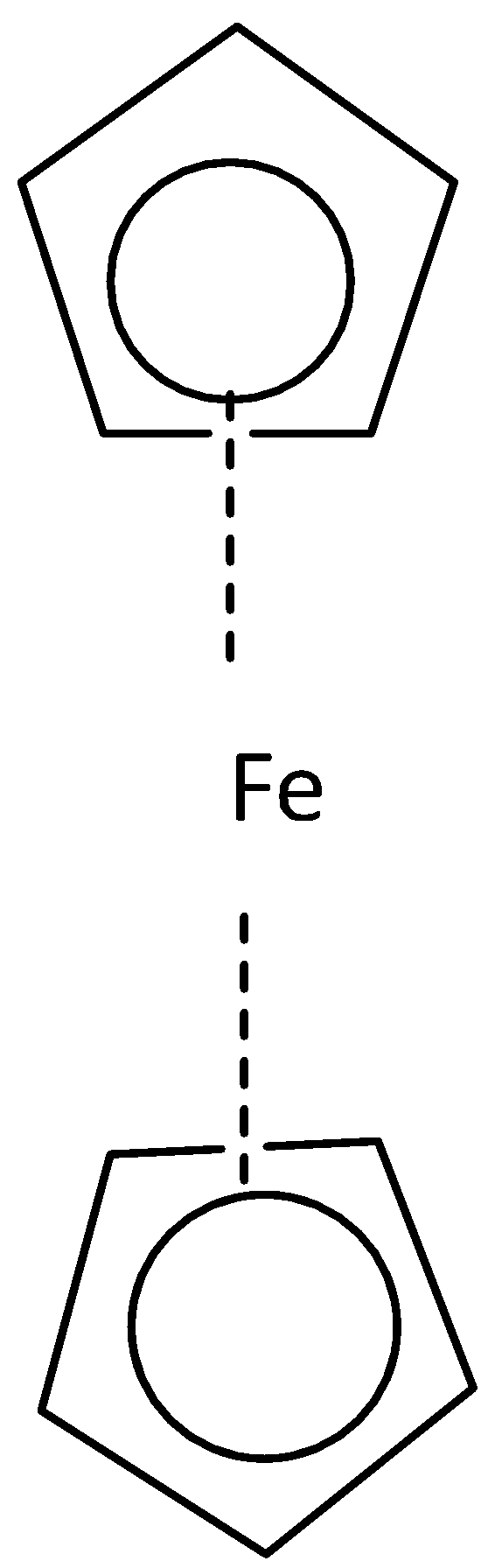
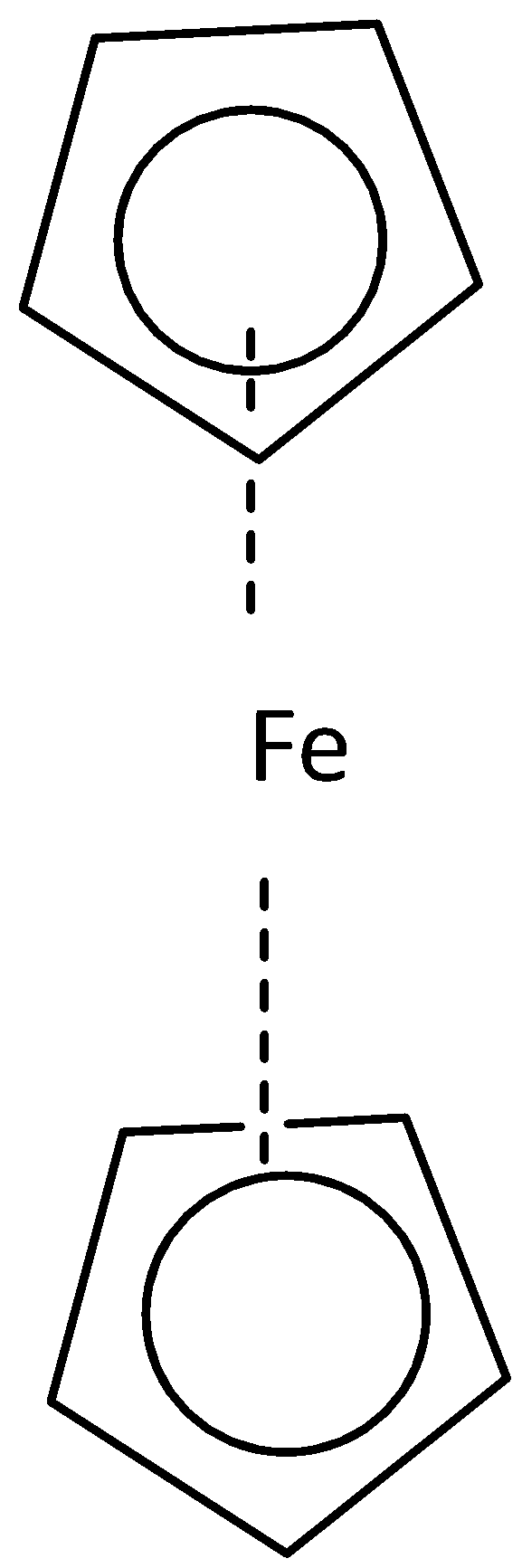
Where both cyclopentadienyl rings are in the opposite direction that is the staggered conformation of ferrocene on the other hand where both the cyclopentadienyl rings in the same direction that is the eclipsed conformation. Point group for staggered conformation of ferrocene is \[{{\text{D}}_{{\text{5d}}}}\] and for eclipsed conformation is \[{{\text{D}}_{{\text{5h}}}}\].
Therefore we can conclude that option D is the correct option, that is \[\left[ {{\text{Fe(}}{{\text{C}}_{\text{5}}}{{\text{H}}_{\text{5}}}{{\text{)}}_{\text{2}}}} \right]\]
Additional information:
Ferrocene undergoes electrophilic substitution reaction faster than benzene indicating that electrons of Cp ring are more readily available. Some electrophilic reactions of ferrocene are given ahead:
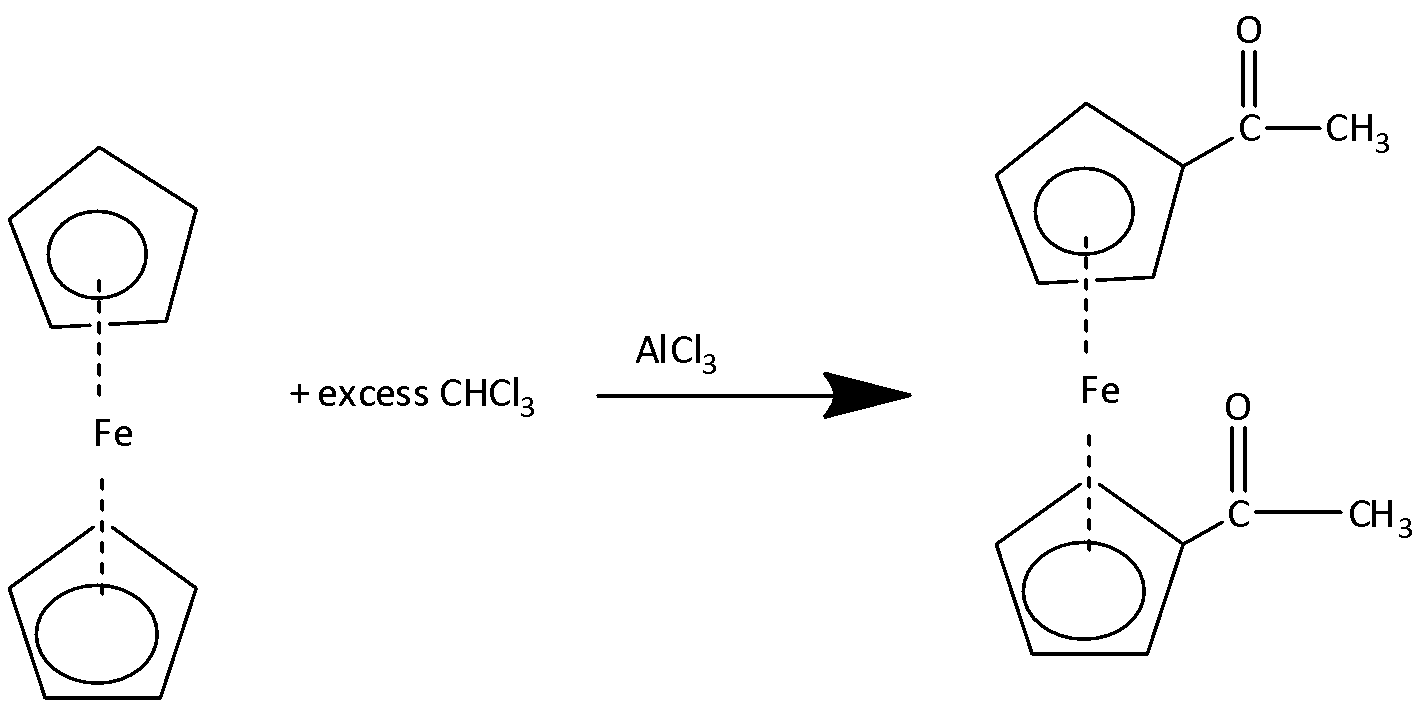
1.Friedel-Craft Acylation:
The reaction of ferrocene with acetyl chloride in the presence of \[{\text{AlC}}{{\text{l}}_{\text{3}}}\] produces mono acetyl derivative of ferrocene.

If the reaction is carried out in the presence of excess amounts of acetyl chloride then diacetyl derivatives of ferrocene are formed.
2,Friedel-Craft Alkylation:
In this reaction we used alkyl halide and \[{\text{AlC}}{{\text{l}}_{\text{3}}}\]at \[{\text{200 atm}}\]to produced mono alkyl derivative of ferrocene. If this reaction is carried out in the presence of excess amounts of alkyl chloride then the dialkyl derivative will be formed.

Note: The cyclopentadiene rings of metallocene are aromatic in nature therefore they do not undergo conjugate diene such as Diels alder reaction. They undergo electrophilic substitution reactions which are characteristic of aromatic compounds.
Complete step by step answer:
In ferrocene \[{\text{Fe}}\] metal is attached with a ligand called cyclopentadiene, the formula for cyclopentadiene is \[{{\text{C}}_{\text{5}}}{{\text{H}}_{\text{5}}}\]. In ferrocene, two cyclopentadienyl ligand present so the formula is \[\left[ {{\text{Fe(}}{{\text{C}}_{\text{5}}}{{\text{H}}_{\text{5}}}{{\text{)}}_{\text{2}}}} \right]\]. Two important conformers of ferrocene are: -


- (a) Staggered conformation (b) eclipsed conformation
Where both cyclopentadienyl rings are in the opposite direction that is the staggered conformation of ferrocene on the other hand where both the cyclopentadienyl rings in the same direction that is the eclipsed conformation. Point group for staggered conformation of ferrocene is \[{{\text{D}}_{{\text{5d}}}}\] and for eclipsed conformation is \[{{\text{D}}_{{\text{5h}}}}\].
Therefore we can conclude that option D is the correct option, that is \[\left[ {{\text{Fe(}}{{\text{C}}_{\text{5}}}{{\text{H}}_{\text{5}}}{{\text{)}}_{\text{2}}}} \right]\]
Additional information:
Ferrocene undergoes electrophilic substitution reaction faster than benzene indicating that electrons of Cp ring are more readily available. Some electrophilic reactions of ferrocene are given ahead:
1.Friedel-Craft Acylation:
The reaction of ferrocene with acetyl chloride in the presence of \[{\text{AlC}}{{\text{l}}_{\text{3}}}\] produces mono acetyl derivative of ferrocene.

If the reaction is carried out in the presence of excess amounts of acetyl chloride then diacetyl derivatives of ferrocene are formed.

2,Friedel-Craft Alkylation:
In this reaction we used alkyl halide and \[{\text{AlC}}{{\text{l}}_{\text{3}}}\]at \[{\text{200 atm}}\]to produced mono alkyl derivative of ferrocene. If this reaction is carried out in the presence of excess amounts of alkyl chloride then the dialkyl derivative will be formed.

Note: The cyclopentadiene rings of metallocene are aromatic in nature therefore they do not undergo conjugate diene such as Diels alder reaction. They undergo electrophilic substitution reactions which are characteristic of aromatic compounds.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




