Explain the structure of diborane .
Answer
597.9k+ views
Hint: Diborane is colourless having chemical formula of ${B_2}{H_6}$.it is toxic in case inhaled. It has an offensive odor. Diborane is formed by boric acid and water. It is easily mixed with air and becomes explosive . so, it must be handled carefully .
Complete answer:
Diborane is made up of a total eight hydrogen atoms and two borons . In which two hydrogen are on the same plane. While the other two hydrogen forms a bridge. Bridge hydrogens are above and below the plane. These bonds are also called banana bonds . The hybridisation of Diborane is $s{p^3}$ in which the length of $B - {H_{bridge}}$ is $1.33$ and length of $B - {H_{ter\min al}}$ is 1.19 $\mathop A\limits^ \circ $.
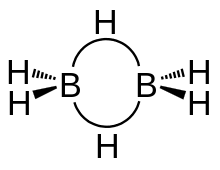
in the diborane the two boron do not form any bonds . The angle between the two terminal hydrogen and one boron is ${120^ \circ }$ and the angle between the bridge hydrogen and
boron is ${97^ \circ }$
Additional information:
Borane can be formed by the reaction of sodium tetrahydridoborate and boron trifluoride etherate . This method is considered the most convenient process for manufacturing diborane .
it can't exist as $B{H_3}$ because boron has three electrons in its valence shell . It is an electron deficient compound and unstable . As after forming a bond with three hydrogens the total number of electrons around the boron is six so it is considered as electron deficient and thus it exists as a dimer which we call diborane .
Note:
Diborane is used in rubber vulcaniser , rocket propellants and a doping agent in the manufacture of semiconductors.It is also used as a reducing agent. It releases a huge amount of energy when burnt in oxygen.
Complete answer:
Diborane is made up of a total eight hydrogen atoms and two borons . In which two hydrogen are on the same plane. While the other two hydrogen forms a bridge. Bridge hydrogens are above and below the plane. These bonds are also called banana bonds . The hybridisation of Diborane is $s{p^3}$ in which the length of $B - {H_{bridge}}$ is $1.33$ and length of $B - {H_{ter\min al}}$ is 1.19 $\mathop A\limits^ \circ $.

in the diborane the two boron do not form any bonds . The angle between the two terminal hydrogen and one boron is ${120^ \circ }$ and the angle between the bridge hydrogen and
boron is ${97^ \circ }$
Additional information:
Borane can be formed by the reaction of sodium tetrahydridoborate and boron trifluoride etherate . This method is considered the most convenient process for manufacturing diborane .
it can't exist as $B{H_3}$ because boron has three electrons in its valence shell . It is an electron deficient compound and unstable . As after forming a bond with three hydrogens the total number of electrons around the boron is six so it is considered as electron deficient and thus it exists as a dimer which we call diborane .
Note:
Diborane is used in rubber vulcaniser , rocket propellants and a doping agent in the manufacture of semiconductors.It is also used as a reducing agent. It releases a huge amount of energy when burnt in oxygen.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




