Explain the mechanism of esterification. Write the reactions involved in dehydration of 1$^o$, 2$^o$, and 3$^o$ alcohols.
Answer
628.5k+ views
Hint: The mechanism of esterification involves the five steps. It is the conversion of carboxylic acids treated with primary alcohols to an ester. The dehydration of alcohols is an elimination reaction including the removal of hydrogen i.e. loose water molecules.
Complete step by step answer:
-Firstly, we will discuss the mechanism of esterification. This involves five steps
Cation formation
-Delocalized carbocation: Carboxyl oxygen gets protonated to give delocalized carbocation, making it a better electrophile.
-Transfer of proton: A proton is transferred to the hydroxyl group to form a good leaving group.
-Formation of pi bond: The hydroxyl group donates a pair of electrons to form a $\pi$ bond.
Ester formation: Deprotonation leads to the ester formation
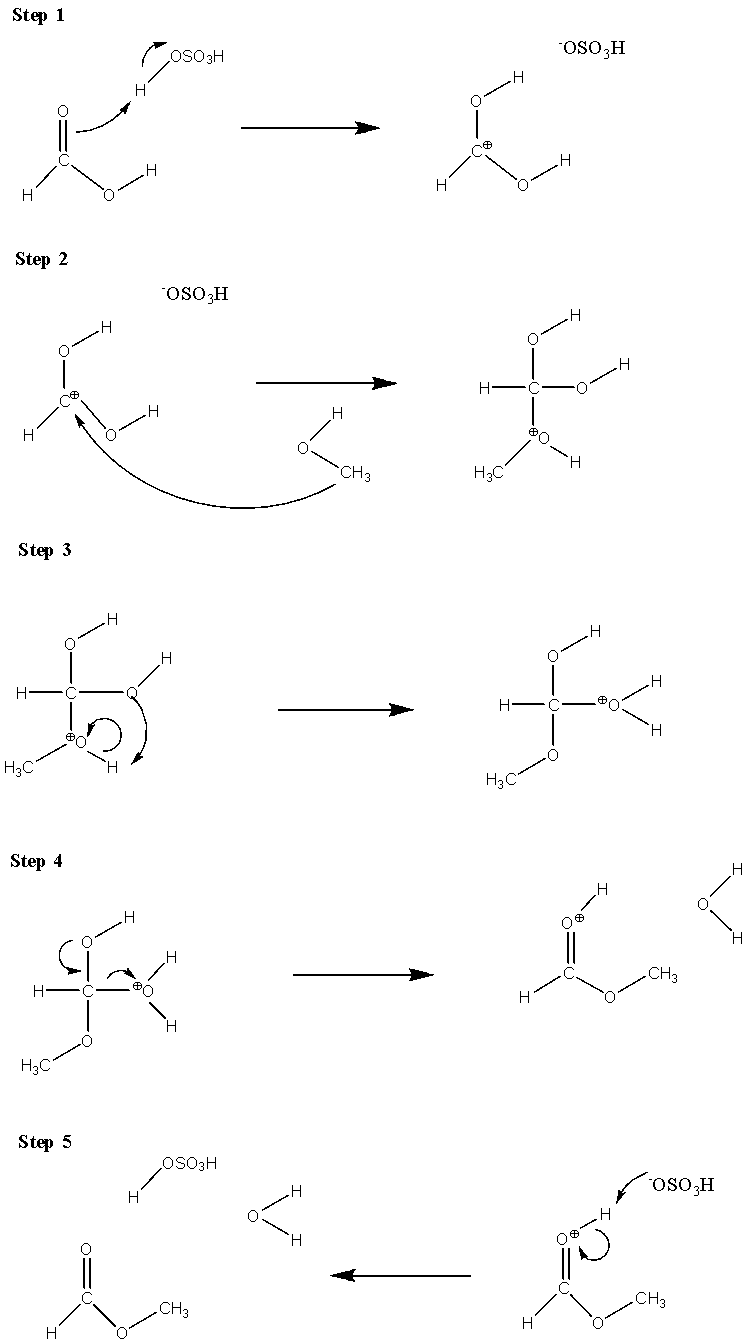
The dehydration of alcohols is done by heating the alcohols in the presence of a strong acid, at high temperatures to form alkenes.
The reaction of dehydration of primary alcohols is:
CH$_3$-CH$_2$-OH $\xrightarrow[\text{443K}]{\text{95%H$_2$SO$_4$}}$ CH$_2$=CH$_2$ +H$_2$O
The reaction of dehydration of secondary alcohols is:
CH$_3$-CH(OH)-CH$_3$ $\xrightarrow[\text{373K}]{\text{60%H$_2$SO$_4$}}$ CH$_3$-CH=CH$_2$ +H$_2$O
The reaction of dehydration of tertiary alcohols is:
CH$_3$-CH(OH)(CH$_3$)-CH$_3$ $\xrightarrow[\text{423K}]{\text{ Al$_2$O$_3$}}$ CH$_3$-C(CH$_3$)=CH$_2$ +H$_2$O
From the above written we get to know the dehydration of alcohols, but there is a difference between three of the reactions, i.e. the temperature required to proceed the reaction.
Thus, in the last we conclude that the esterification leads to ester formation, and dehydration of alcohols to alkenes.
Note:
Don’t get confused between the esterification, and the dehydration of alcohols. The end product in both reactions is different, but the similarity is involvement of alcohols. The dehydration of alcohol involves various conditions; the end product is alkene by the removal of water.
Complete step by step answer:
-Firstly, we will discuss the mechanism of esterification. This involves five steps
Cation formation
-Delocalized carbocation: Carboxyl oxygen gets protonated to give delocalized carbocation, making it a better electrophile.
-Transfer of proton: A proton is transferred to the hydroxyl group to form a good leaving group.
-Formation of pi bond: The hydroxyl group donates a pair of electrons to form a $\pi$ bond.
Ester formation: Deprotonation leads to the ester formation

The dehydration of alcohols is done by heating the alcohols in the presence of a strong acid, at high temperatures to form alkenes.
The reaction of dehydration of primary alcohols is:
CH$_3$-CH$_2$-OH $\xrightarrow[\text{443K}]{\text{95%H$_2$SO$_4$}}$ CH$_2$=CH$_2$ +H$_2$O
The reaction of dehydration of secondary alcohols is:
CH$_3$-CH(OH)-CH$_3$ $\xrightarrow[\text{373K}]{\text{60%H$_2$SO$_4$}}$ CH$_3$-CH=CH$_2$ +H$_2$O
The reaction of dehydration of tertiary alcohols is:
CH$_3$-CH(OH)(CH$_3$)-CH$_3$ $\xrightarrow[\text{423K}]{\text{ Al$_2$O$_3$}}$ CH$_3$-C(CH$_3$)=CH$_2$ +H$_2$O
From the above written we get to know the dehydration of alcohols, but there is a difference between three of the reactions, i.e. the temperature required to proceed the reaction.
Thus, in the last we conclude that the esterification leads to ester formation, and dehydration of alcohols to alkenes.
Note:
Don’t get confused between the esterification, and the dehydration of alcohols. The end product in both reactions is different, but the similarity is involvement of alcohols. The dehydration of alcohol involves various conditions; the end product is alkene by the removal of water.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




