During conductometric titration of \[0.1M\] $HCl$ with $1.0M$ $KOH$ , which of the following will be observed?
A. Resistance of the solution decreases up to equivalence point and then increases
B. Resistance of the solution increases up to equivalence point and then decreases
C. Conductance increases up to equivalence point and then decreases
D. Conductance decreases up to equivalence point and then becomes almost constant
Answer
590.1k+ views
Hint: Conductometric titration is defined as the technique to monitor electrolytic conductivity by adding one reactant in a mixture. You should also have an idea about resistance. Resistance is defined as the obstruction to current flow and how resistance and conductance vary with the changing concentration of ions.
Complete step by step answer:
Conductometric titration is defined as a standard technique in which the electrolytic conductivity of the reaction mixture is monitored continuously when one reactant is added in the mixture. When the conductivity shows sudden change, that point is known as the equivalence point. If there is a change in the concentration of ${{H}^{+}}$ ions or $O{{H}^{-}}$ ions, the conductivity will automatically increase or decrease. This method is commonly used for colored solutions which cannot be used with normal indicators.
During this titration, the conductance of the solution decreases first, then reaches the end point, that is the minimum value at equivalence point and then the conductance increases. In this question, strong acid and strong base is used which have the ability to dissociate the ions completely. After the equivalence point the resistance decreases due to the increase in the hydroxyl ions in the solution.
As we know, resistance is inversely proportional to conductance. Therefore, the conductance increases first and reaches the equivalent point and then the conductance decreases.
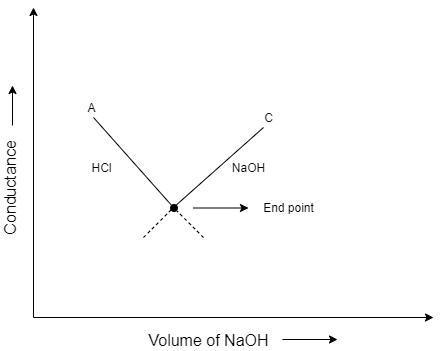
So, the correct answer is Option B.
Note: Resistance is defined as the measure of the obstruction to current flow. It is denoted by a symbol omega $(\Omega )$. Its SI unit is $ohms$ . If the resistance is high, then the current flow is low and if the resistance is low, then the current flow is high.
Conductance is defined as a measure of the ease with which the current flows through a material. If the electrons are flowing easily, that means conductance is high and vice-versa.
Conductance is inversely proportional to resistance. When a reactant is added to the solution, it changes the conductivity of the solution. Then you will see a sudden change that gives an end point.
Complete step by step answer:
Conductometric titration is defined as a standard technique in which the electrolytic conductivity of the reaction mixture is monitored continuously when one reactant is added in the mixture. When the conductivity shows sudden change, that point is known as the equivalence point. If there is a change in the concentration of ${{H}^{+}}$ ions or $O{{H}^{-}}$ ions, the conductivity will automatically increase or decrease. This method is commonly used for colored solutions which cannot be used with normal indicators.
During this titration, the conductance of the solution decreases first, then reaches the end point, that is the minimum value at equivalence point and then the conductance increases. In this question, strong acid and strong base is used which have the ability to dissociate the ions completely. After the equivalence point the resistance decreases due to the increase in the hydroxyl ions in the solution.
As we know, resistance is inversely proportional to conductance. Therefore, the conductance increases first and reaches the equivalent point and then the conductance decreases.

So, the correct answer is Option B.
Note: Resistance is defined as the measure of the obstruction to current flow. It is denoted by a symbol omega $(\Omega )$. Its SI unit is $ohms$ . If the resistance is high, then the current flow is low and if the resistance is low, then the current flow is high.
Conductance is defined as a measure of the ease with which the current flows through a material. If the electrons are flowing easily, that means conductance is high and vice-versa.
Conductance is inversely proportional to resistance. When a reactant is added to the solution, it changes the conductivity of the solution. Then you will see a sudden change that gives an end point.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Discuss the various forms of bacteria class 11 biology CBSE




