How can I draw the Lewis structure for ${{H}_{2}}O$ ?
Answer
599.1k+ views
Hint Lewis dot structures also known as electron dot structures. Lewis dot structures represent the valence electrons of the atoms present within the given molecule. By Lewis dot structures we can visualize the bonding electrons and lone pair of electrons present in the molecule.
Complete answer:
- In the question it is given that to draw the Lewis dot structures of the water molecule.
- We know that the atomic number of oxygen is 8 and has eight electrons in its electronic configuration.
- The electronic configuration of oxygen is $1{{s}^{2}}2{{s}^{2}}2{{p}^{4}}$
- So oxygen has six valence electrons in 2s and 2p orbital.
- The Lewis dot structure of the beryllium is as follows.
- Coming to hydrogen, we know that the hydrogen element has one valence electron in its electronic configuration.
- The Lewis dot structure of hydrogen is as follows.
- Now by taking the above structures as examples we can draw the Lewis dot structure of ${{H}_{2}}O$ and it is as follows.
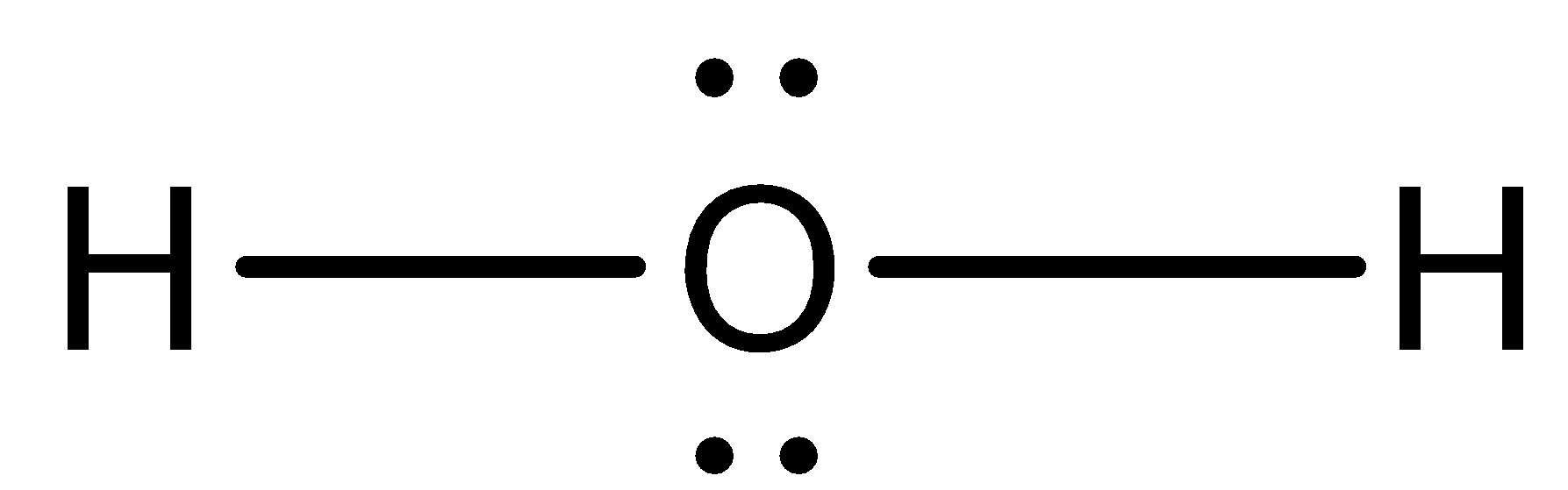
- From the above Lewis dot structure we can easily say that an oxygen atom has two lone pairs of electrons and hydrogen has zero lone pair of electrons in water molecules.
Note: By using Lewis dot structures of the molecules we can determine the formation of the chemical bonds in various molecules. We can draw the Lewis dot structures for the compounds containing covalent bonds and also we can draw for coordination compounds.
Complete answer:
- In the question it is given that to draw the Lewis dot structures of the water molecule.
- We know that the atomic number of oxygen is 8 and has eight electrons in its electronic configuration.
- The electronic configuration of oxygen is $1{{s}^{2}}2{{s}^{2}}2{{p}^{4}}$
- So oxygen has six valence electrons in 2s and 2p orbital.
- The Lewis dot structure of the beryllium is as follows.
- Coming to hydrogen, we know that the hydrogen element has one valence electron in its electronic configuration.
- The Lewis dot structure of hydrogen is as follows.
- Now by taking the above structures as examples we can draw the Lewis dot structure of ${{H}_{2}}O$ and it is as follows.

- From the above Lewis dot structure we can easily say that an oxygen atom has two lone pairs of electrons and hydrogen has zero lone pair of electrons in water molecules.
Note: By using Lewis dot structures of the molecules we can determine the formation of the chemical bonds in various molecules. We can draw the Lewis dot structures for the compounds containing covalent bonds and also we can draw for coordination compounds.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




