Draw diagram of an experimental arrangement for observing scattering of light in
colloidal solution. Name the two chemicals used in this activity.
Answer
588.3k+ views
Hint:The arrangement used for the scattering of light consists of a source of light placed in front of the converging lens. A transparent glass tank, a cardboard that contains hole, a converging lens that converges the light on the screen. The chemicals used in this activity will be used for producing Tyndall effect.
COMPLETE STEP BY STEP ANSWER:
The diagram showing the scattering of light in the colloidal solution is given by
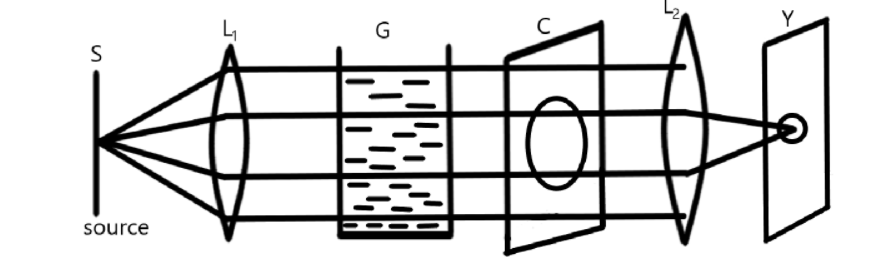
Here, $S$ is the source of light from the rays of light are emerging, ${L_1}$ and ${L_2}$ are the convex lenses, $G$ is the transparent glass surface that contains colloidal solution, $C$ is the cardboard that contains hole and $Y$ is the screen on which we get an image.
As shown in the figure, a source $S$ is placed in front of a convex lens that allows the rays of light to fall in the transparent glass tank that contains the colloidal solution that enables the Tyndall effect of the rays of light. After the Tyndall effect, these rays of light fall on the cardboard that consists of hole.
Some of the rays of light pass through the holes and fall on the converging lens. This converging lens will converge the rays of light on the screen that forms the image on the screen.
The solution in the tank contains $2l$ of the water in which $1\,ml$ to $2\,ml$ of sulphuric acid
$\left( {{H_2}S{O_4}} \right)$ is added.
Therefore, the two chemicals used in this activity are water and sulphuric acid.
NOTE:The above arrangement is used to show the scattering of light. This arrangement can also be used to determine why the sky appears blue and why sunset appears red. When the wavelength of the light becomes lower, the color will be blue. On the other hand, when the wavelength of the light will be greater, the color will be red.
COMPLETE STEP BY STEP ANSWER:
The diagram showing the scattering of light in the colloidal solution is given by

Here, $S$ is the source of light from the rays of light are emerging, ${L_1}$ and ${L_2}$ are the convex lenses, $G$ is the transparent glass surface that contains colloidal solution, $C$ is the cardboard that contains hole and $Y$ is the screen on which we get an image.
As shown in the figure, a source $S$ is placed in front of a convex lens that allows the rays of light to fall in the transparent glass tank that contains the colloidal solution that enables the Tyndall effect of the rays of light. After the Tyndall effect, these rays of light fall on the cardboard that consists of hole.
Some of the rays of light pass through the holes and fall on the converging lens. This converging lens will converge the rays of light on the screen that forms the image on the screen.
The solution in the tank contains $2l$ of the water in which $1\,ml$ to $2\,ml$ of sulphuric acid
$\left( {{H_2}S{O_4}} \right)$ is added.
Therefore, the two chemicals used in this activity are water and sulphuric acid.
NOTE:The above arrangement is used to show the scattering of light. This arrangement can also be used to determine why the sky appears blue and why sunset appears red. When the wavelength of the light becomes lower, the color will be blue. On the other hand, when the wavelength of the light will be greater, the color will be red.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Class 11 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Accountancy: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

In what year Guru Nanak Dev ji was born A15 April 1469 class 11 social science CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE




