Define an atomic orbital.
Answer
598.5k+ views
Hint: An atom is the smallest unit of matter which forms a chemical element. Every solid, liquid, gas consists of atoms. Atoms consist of protons, neutrons and electrons.
Complete answer:
Atomic orbitals are the 3-dimensional spaces around the nucleus where the probability of finding an electron is maximum. The molecules orbitals are formed by combining the atomic orbitals. In quantum chemistry we have encounter orbitals which are s, p, d and f subshells. Orbitals are of different shapes and sizes and they can be determined by the square of the wave function.
Shapes of orbitals s, p, d and f are given below:
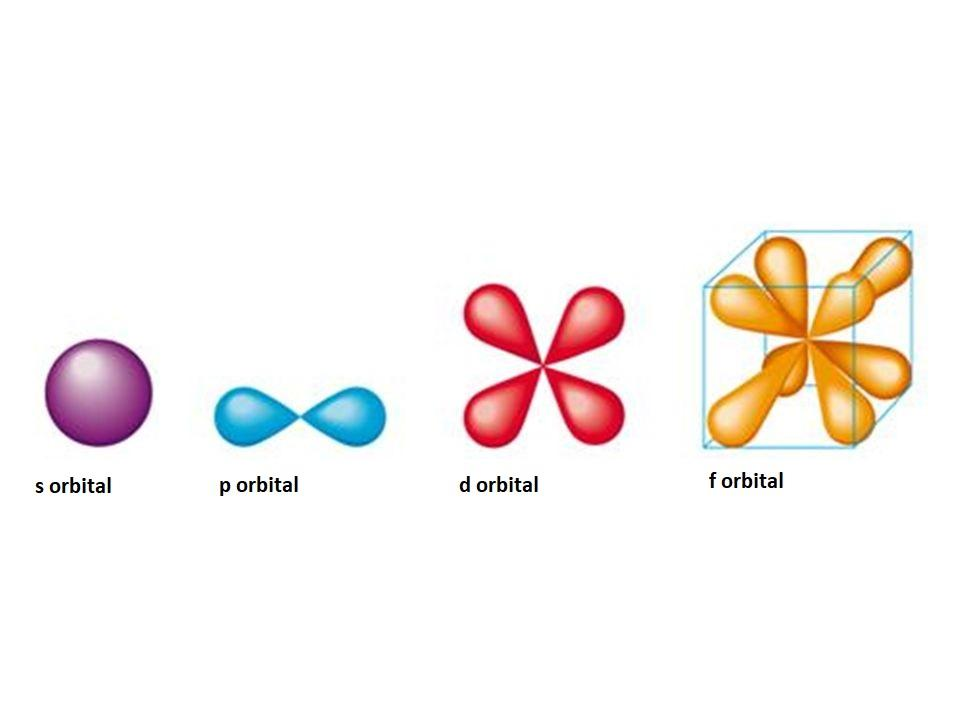
Orbitals are of different shapes the s orbital has a spherical shape, the p orbital has a dumbbell shape the d orbital has a double dumbbell shape. The maximum number of electrons which can be present in an orbital is two. Almost all the orbitals are directional in nature except the s orbital, it is non directional in nature. The concept of orbitals is explained by the Heisenberg Uncertainty principle.
Note:
Do not get confused between orbit and orbitals. Orbit is a well-defined circular path around the nucleus in which the electrons revolve whereas atomic orbitals are the 3-dimensional spaced around the nucleus where the probability of finding an electron is maximum.
Orbits represent the motion of electrons in a plane and they are non-directional in nature. The orbits are either circular or elliptical in shape. The nodal plane is a plane where the probability of finding an electron is zero.
Complete answer:
Atomic orbitals are the 3-dimensional spaces around the nucleus where the probability of finding an electron is maximum. The molecules orbitals are formed by combining the atomic orbitals. In quantum chemistry we have encounter orbitals which are s, p, d and f subshells. Orbitals are of different shapes and sizes and they can be determined by the square of the wave function.
Shapes of orbitals s, p, d and f are given below:

Orbitals are of different shapes the s orbital has a spherical shape, the p orbital has a dumbbell shape the d orbital has a double dumbbell shape. The maximum number of electrons which can be present in an orbital is two. Almost all the orbitals are directional in nature except the s orbital, it is non directional in nature. The concept of orbitals is explained by the Heisenberg Uncertainty principle.
Note:
Do not get confused between orbit and orbitals. Orbit is a well-defined circular path around the nucleus in which the electrons revolve whereas atomic orbitals are the 3-dimensional spaced around the nucleus where the probability of finding an electron is maximum.
Orbits represent the motion of electrons in a plane and they are non-directional in nature. The orbits are either circular or elliptical in shape. The nodal plane is a plane where the probability of finding an electron is zero.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




