Consider the reaction,\[{\text{C}}{{\text{H}}_{\text{3}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{Br + NaCN }} \to {\text{ C}}{{\text{H}}_{\text{3}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{CN + NaBr }}\].
This reaction will be the fastest in:
A. N,N’-dimethylformamide(DMF)
B. Water
C. ethanol
D. methanol
Answer
596.1k+ views
Hint:The given reaction is a nucleophilic substitution \[{\text{(S}}{{\text{N}}_{\text{2}}}{\text{)}}\] reaction. The rate of nucleophilic substitution reaction depends on the solvent. Nucleophilic substitution reactions are favourable in a polar aprotic solvent.
Complete answer:
The reaction given to us is:
\[{\text{C}}{{\text{H}}_{\text{3}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{Br + NaCN }} \to {\text{ C}}{{\text{H}}_{\text{3}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{CN + NaBr }}\]
Here we can see that there is a substitution of the bromide group by the cyanide group. Since given alkyl halide is primary alkyl halide so we can say that it is \[{\text{S}}{{\text{N}}_{\text{2}}}\] a reaction. The rate of nucleophilic substitution reaction depends on the solvent. As we know there are two types of solvents.
1. Polar protic solvent
2. Polar aprotic solvent.
Now, we will determine the type of solvent given to us as follows:
A. The structure of N,N-dimethylformamide(DMF) is :
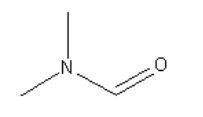
So, DMF is a polar aprotic solvent as the hydrogen atom is not bonded to electronegative nitrogen and oxygen atoms.
B. The structure of water is
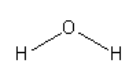
So, water is a polar protic solvent.
C. The structure of ethanol is

So, ethanol is a polar protic solvent.
D. The structure of methanol is
\[{\text{C}}{{\text{H}}_{\text{3}}}{\text{ - OH}}\]
So, methanol is a polar protic solvent.
Polar protic solvent tends to form a hydrogen bond with a nucleophile and hinder the attack of the nucleophile. While the polar aprotic solvent does not form a hydrogen bond with a nucleophile. Thus, the polar aprotic solvent does not hinder the nucleophile so the rate of nucleophilic substitution is faster in the polar aprotic solvent.
Out of the given solvent to us only of N,N’-dimethylformamide(DMF) is polar aprotic solvent rest all (water, ethanol and methanol) are polar protic solvent. So, the given reaction is fastest in N,N’-dimethylformamide(DMF).
Thus, the correct option is (A) of N, N’-dimethylformamide (DMF).
Note:
In \[{\text{S}}{{\text{N}}_{\text{2}}}\] reaction attack of nucleophiles is the rate-determining step. So the solvent that favours the attack of nucleophile is the favourable solvent. As polar aprotic solvent is a non-hindering solvent it speeds up the reaction.
Complete answer:
The reaction given to us is:
\[{\text{C}}{{\text{H}}_{\text{3}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{Br + NaCN }} \to {\text{ C}}{{\text{H}}_{\text{3}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{CN + NaBr }}\]
Here we can see that there is a substitution of the bromide group by the cyanide group. Since given alkyl halide is primary alkyl halide so we can say that it is \[{\text{S}}{{\text{N}}_{\text{2}}}\] a reaction. The rate of nucleophilic substitution reaction depends on the solvent. As we know there are two types of solvents.
1. Polar protic solvent
2. Polar aprotic solvent.
Now, we will determine the type of solvent given to us as follows:
A. The structure of N,N-dimethylformamide(DMF) is :

So, DMF is a polar aprotic solvent as the hydrogen atom is not bonded to electronegative nitrogen and oxygen atoms.
B. The structure of water is

So, water is a polar protic solvent.
C. The structure of ethanol is

So, ethanol is a polar protic solvent.
D. The structure of methanol is
\[{\text{C}}{{\text{H}}_{\text{3}}}{\text{ - OH}}\]
So, methanol is a polar protic solvent.
Polar protic solvent tends to form a hydrogen bond with a nucleophile and hinder the attack of the nucleophile. While the polar aprotic solvent does not form a hydrogen bond with a nucleophile. Thus, the polar aprotic solvent does not hinder the nucleophile so the rate of nucleophilic substitution is faster in the polar aprotic solvent.
Out of the given solvent to us only of N,N’-dimethylformamide(DMF) is polar aprotic solvent rest all (water, ethanol and methanol) are polar protic solvent. So, the given reaction is fastest in N,N’-dimethylformamide(DMF).
Thus, the correct option is (A) of N, N’-dimethylformamide (DMF).
Note:
In \[{\text{S}}{{\text{N}}_{\text{2}}}\] reaction attack of nucleophiles is the rate-determining step. So the solvent that favours the attack of nucleophile is the favourable solvent. As polar aprotic solvent is a non-hindering solvent it speeds up the reaction.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




