$C{{O}_{2}}$ and $Si{{O}_{2}}$ is a solid or gas?
(A) $C{{O}_{2}}$ gas and $Si{{O}_{2}}$ solid
(B) $C{{O}_{2}}$ solid and $Si{{O}_{2}}$ gas
(C) $C{{O}_{2}}$ gas and $Si{{O}_{2}}$ gas
Answer
606.3k+ views
Hint: Both carbon and silicon belong to the same group in the periodic table and this means that these atoms have similar properties. $C{{O}_{2}}$ consists of a carbon atom which is covalently double bonded to two oxygen atoms and $Si{{O}_{2}}$ or silicon dioxide is an oxide of silicon.
Complete answer:
-As we know, both carbon and silicon belong to the same group in the periodic table. Hence, both these atoms should have similar properties. These atoms have a tendency to show catenation which is the property of an element with which it can form a long chain by linking with other atoms of the same element.
- In carbon dioxide there is a double bond between the oxygen and carbon atoms. (O=C=O), each molecule is attracted to the other molecules through the London forces or van der waals forces.
- Because of the small atomic size, carbon atom possesses partial triple bond character with the neighboring oxygen atoms and due to the linear structure $C{{O}_{2}}$ is non-polar and hence they exhibit weak van der waals forces. As a result, Carbon dioxide ($C{{O}_{2}}$ ) exists as gas.
- In the case of $Si{{O}_{2}}$, every silicon atom is covalently bonded to four oxygen atoms and every oxygen atom is bonded to two silicon atoms. This will result in the formation of a giant tetrahedral structure which is shown below
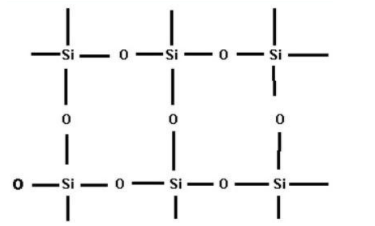
Because of this giant network, the bonding in $Si{{O}_{2}}$ is very strong and thus it exists as a solid. Therefore the $C{{O}_{2}}$ molecule exists as gas and $Si{{O}_{2}}$ exists as solid.
Thus the answer is option (A) $C{{O}_{2}}$ gas and $Si{{O}_{2}}$ solid.
Note: It should be noted that, $C{{O}_{2}}$ is an acidic oxide (forms carbonic acid on reaction with water) and $Si{{O}_{2}}$ doesn’t react with water, due to the difficulty of breaking up the giant covalent structure. Instead, $Si{{O}_{2}}$ is very weakly acidic when reacting with bases.
Complete answer:
-As we know, both carbon and silicon belong to the same group in the periodic table. Hence, both these atoms should have similar properties. These atoms have a tendency to show catenation which is the property of an element with which it can form a long chain by linking with other atoms of the same element.
- In carbon dioxide there is a double bond between the oxygen and carbon atoms. (O=C=O), each molecule is attracted to the other molecules through the London forces or van der waals forces.
- Because of the small atomic size, carbon atom possesses partial triple bond character with the neighboring oxygen atoms and due to the linear structure $C{{O}_{2}}$ is non-polar and hence they exhibit weak van der waals forces. As a result, Carbon dioxide ($C{{O}_{2}}$ ) exists as gas.
- In the case of $Si{{O}_{2}}$, every silicon atom is covalently bonded to four oxygen atoms and every oxygen atom is bonded to two silicon atoms. This will result in the formation of a giant tetrahedral structure which is shown below

Because of this giant network, the bonding in $Si{{O}_{2}}$ is very strong and thus it exists as a solid. Therefore the $C{{O}_{2}}$ molecule exists as gas and $Si{{O}_{2}}$ exists as solid.
Thus the answer is option (A) $C{{O}_{2}}$ gas and $Si{{O}_{2}}$ solid.
Note: It should be noted that, $C{{O}_{2}}$ is an acidic oxide (forms carbonic acid on reaction with water) and $Si{{O}_{2}}$ doesn’t react with water, due to the difficulty of breaking up the giant covalent structure. Instead, $Si{{O}_{2}}$ is very weakly acidic when reacting with bases.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




