Classify $Na{{H}_{2}}P{{O}_{2}}$ and $NaHS{{O}_{4}}$ salts as acid salt or normal salt.
Answer
603.3k+ views
Hint: In oxoacids, the hydrogen atom that is attached to the oxygen atom causes the acidic nature of the oxoacids. So if the compound has more than one acidic hydrogen atom then the resulting salt will be acidic, and if the compound has only one acidic hydrogen atom then the resulting salt is normal or neutral.
Complete step by step answer:
-Both compounds given in the question are salts of oxoacids of respective elements. In $Na{{H}_{2}}P{{O}_{2}}$, phosphorus is the central atom, so it must be salt of oxoacids of phosphorus. In $NaHS{{O}_{4}}$, sulfur is the central atom, so it must be salt of oxoacids of sulfur.
-In oxoacids, the hydrogen atom that is attached to the oxygen atom causes the acidic nature of the oxoacids. So if the compound has more than one acidic hydrogen atom then the resulting salt will be acidic, and if the compound has only one acidic hydrogen atom then the resulting salt is normal or neutral.
-So the $Na{{H}_{2}}P{{O}_{2}}$, is the salt of ${{H}_{3}}P{{O}_{2}}$, there is only one acidic hydrogen atom is present as you can see in the diagram:
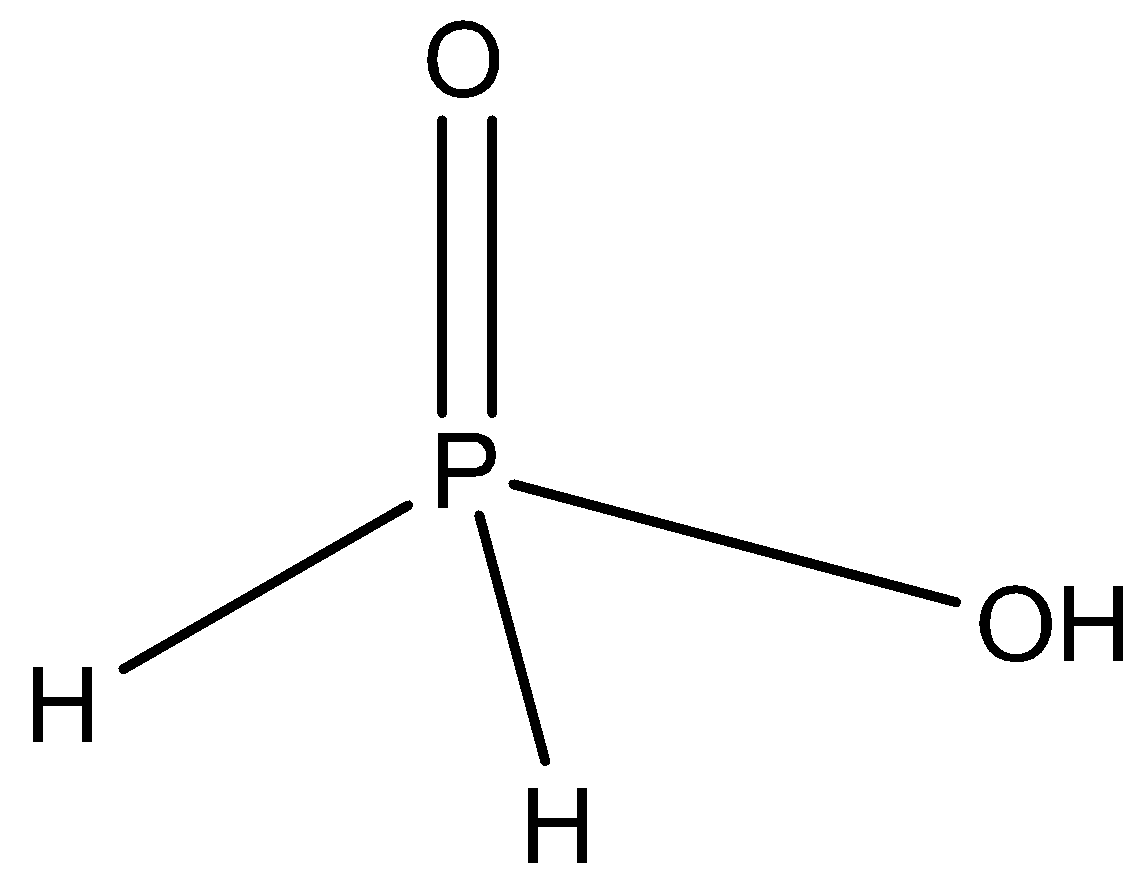
-So this hydrogen is substituted with sodium metal, so it is a normal or neutral salt.
-The $NaHS{{O}_{4}}$, is the salt of ${{H}_{2}}S{{O}_{4}}$, in ${{H}_{2}}S{{O}_{4}}$ there are two acidic hydrogen atoms are present as you can see in the diagram:
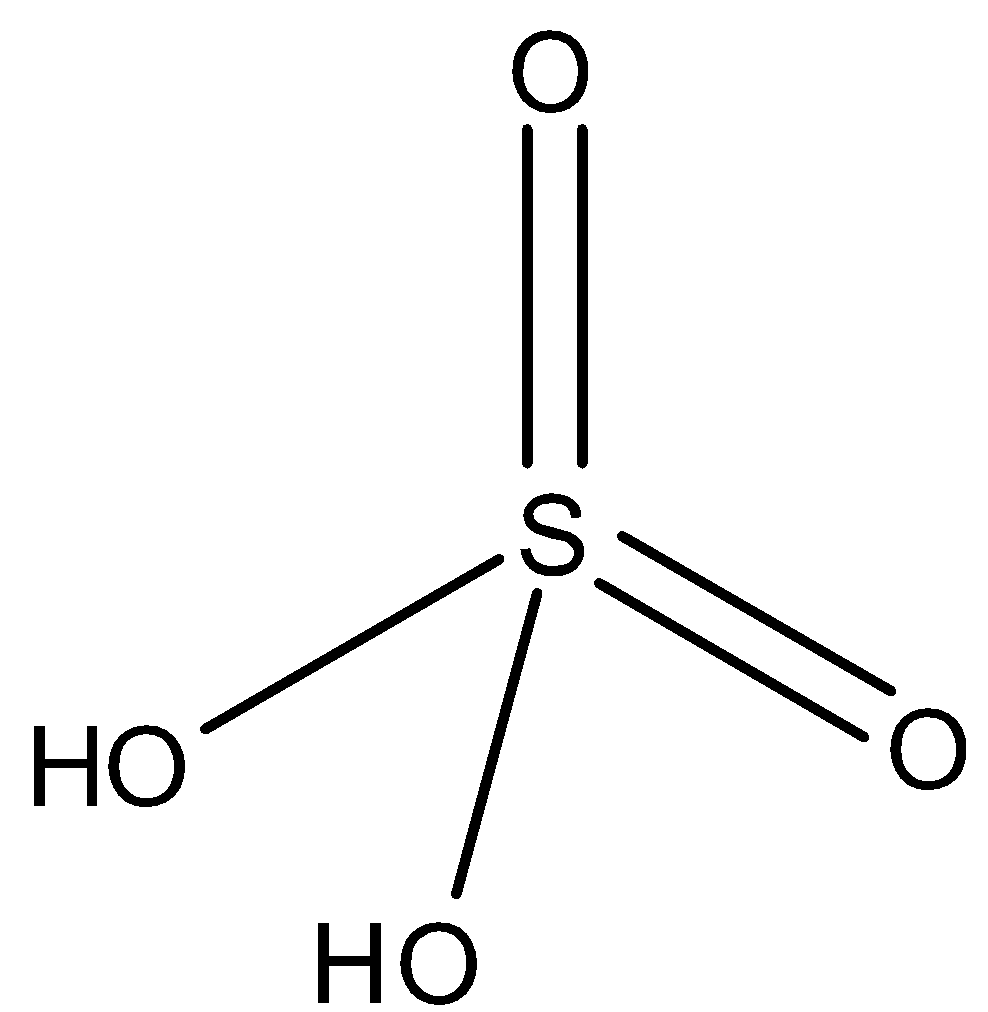
-So one hydrogen atom is substituted with sodium metal or partial substitution takes place, so it is an acid salt.
Note: The pH of the neutral salt or normal salt ranges from 5-7 and does not show any acidic or basic nature. Acid salt is always formed by incomplete replacement of hydrogen atoms.
Complete step by step answer:
-Both compounds given in the question are salts of oxoacids of respective elements. In $Na{{H}_{2}}P{{O}_{2}}$, phosphorus is the central atom, so it must be salt of oxoacids of phosphorus. In $NaHS{{O}_{4}}$, sulfur is the central atom, so it must be salt of oxoacids of sulfur.
-In oxoacids, the hydrogen atom that is attached to the oxygen atom causes the acidic nature of the oxoacids. So if the compound has more than one acidic hydrogen atom then the resulting salt will be acidic, and if the compound has only one acidic hydrogen atom then the resulting salt is normal or neutral.
-So the $Na{{H}_{2}}P{{O}_{2}}$, is the salt of ${{H}_{3}}P{{O}_{2}}$, there is only one acidic hydrogen atom is present as you can see in the diagram:

-So this hydrogen is substituted with sodium metal, so it is a normal or neutral salt.
-The $NaHS{{O}_{4}}$, is the salt of ${{H}_{2}}S{{O}_{4}}$, in ${{H}_{2}}S{{O}_{4}}$ there are two acidic hydrogen atoms are present as you can see in the diagram:

-So one hydrogen atom is substituted with sodium metal or partial substitution takes place, so it is an acid salt.
Note: The pH of the neutral salt or normal salt ranges from 5-7 and does not show any acidic or basic nature. Acid salt is always formed by incomplete replacement of hydrogen atoms.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




