
Choose the correct statement(s)
(i) \[2 - \] Methylpropanoic acid & ethanol react in the presence of conc. \[{H_2}S{O_4}\] to give a compound with a fruity smell.
(ii) In\[{\left[ {Ni{{\left( {CN} \right)}_4}} \right]^{2 - }}\], the \[d - \]orbital involved in the hybridization is \[{d_{{x^2} - {y^2}}}\]
(iii) Solid \[{N_2}{O_5}\] contains discrete units of \[N{O_2}^ + \] and\[N{O_3}^ - \] .
A. I, II
B. II, III
C. I, II, III
D. I, III
Answer
552k+ views
Hint: The first statement belongs to the esterification reaction between an acid and an alcohol. The valence bond theory is used to determine the coordination complex structure for the second statement. The third statement belongs to the existence of \[{N_2}{O_5}\] in solid phase.
Complete step by step answer:
Let us check the correctness of the given statements one by one.
(i) \[2 - \] Methylpropanoic acid & ethanol react in the presence of conc. \[{H_2}S{O_4}\] to give a compound with a fruity smell.
The given reaction is an organic chemical reaction known as Fischer esterification. In this reaction, a carboxylic acid is treated with an alcohol in the presence of catalytic acid and leads to formation of an ester molecule and water. For the given substrates the \[2 - \] methylpropanoic acid or isobutyric acid is the carboxylic acid and the ethanol is the alcohol which is reacted in presence on catalytic \[{H_2}S{O_4}\]. The product of the reaction is ethyl isobutyrate which has a fruity smell. The reaction is
$C{H_3}CH(C{H_3})COOH + {C_2}{H_5}OH\xrightarrow{{{H_2}S{O_4}}}C{H_3}CH(C{H_3})COO{C_2}{H_5} + {H_2}O$
Hence the given statement is correct.
(ii) In \[{\left[ {Ni{{\left( {CN} \right)}_4}} \right]^{2 - }}\], the \[d - \]orbital involved in the hybridization is \[{d_{{x^2} - {y^2}}}\].
In order to find the \[d - \] orbital involved, the hybridization of the given complex is to be determined. The given complex is a tetrahedral complex of nickel. Let the oxidation state of the central atom nickel is x. It is an anionic complex with two negative charges.
$x + 4( - 1) = - 2$
$x = + 2$.
Nickel is an element in the periodic table with atomic number \[28\] and electronic configuration \[\left[ {Ar} \right]3{d^8}4{s^2}\] . The electronic configuration of \[N{i^{2 + }}\] is\[\left[ {Ar} \right]3{d^8}\] . Thus the four cyanide atoms occupy the one \[d\], one \[s\] and two \[p\]-orbitals of the \[N{i^{2 + }}\] ion.
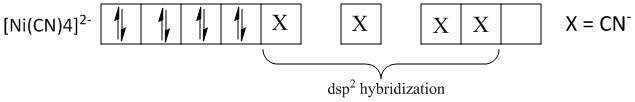
The \[{d_{{x^2} - {y^2}}}\] orbital is placed in the xy plane with the lobes directed along the x and y axes. Thus the \[ds{p^2}\] hybridization favors a planar geometry where all the four hybridized orbitals lie along the xy plane. Thus \[{d_{{x^2} - {y^2}}}\] orbital is used for \[ds{p^2}\] hybridization and the statement is correct.
(iii) Solid \[{N_2}{O_5}\] contains discrete units of \[N{O_2}^ + \] and \[N{O_3}^ - \].
\[{N_2}{O_5}\] is known as Dinitrogen pentoxide. The compound is a binary nitrogen oxide, which consists of nitrogen and oxygen. It exists as colourless crystals with melting point of \[41\;^\circ C\] and boiling point of \[47\;^\circ C\].
\[{N_2}{O_5}\] exists in the form of two structures depending on the phases of the compound. In the solid phase it is a salt called nitronium nitrate. It consists of separate nitronium cations \[{\left[ {N{O_2}} \right]^ + }\] and nitrate anions \[{\left[ {N{O_3}} \right]^ - }\] . The compound in the gas phase exists as a covalently bound molecule.
So, the correct answer is Option C.
Note: The concept of hybridization is important for determining the structure of the complex. The choice of \[d - \] orbitals depends on the type of hybridization and the geometry of the molecule.
Complete step by step answer:
Let us check the correctness of the given statements one by one.
(i) \[2 - \] Methylpropanoic acid & ethanol react in the presence of conc. \[{H_2}S{O_4}\] to give a compound with a fruity smell.
The given reaction is an organic chemical reaction known as Fischer esterification. In this reaction, a carboxylic acid is treated with an alcohol in the presence of catalytic acid and leads to formation of an ester molecule and water. For the given substrates the \[2 - \] methylpropanoic acid or isobutyric acid is the carboxylic acid and the ethanol is the alcohol which is reacted in presence on catalytic \[{H_2}S{O_4}\]. The product of the reaction is ethyl isobutyrate which has a fruity smell. The reaction is
$C{H_3}CH(C{H_3})COOH + {C_2}{H_5}OH\xrightarrow{{{H_2}S{O_4}}}C{H_3}CH(C{H_3})COO{C_2}{H_5} + {H_2}O$
Hence the given statement is correct.
(ii) In \[{\left[ {Ni{{\left( {CN} \right)}_4}} \right]^{2 - }}\], the \[d - \]orbital involved in the hybridization is \[{d_{{x^2} - {y^2}}}\].
In order to find the \[d - \] orbital involved, the hybridization of the given complex is to be determined. The given complex is a tetrahedral complex of nickel. Let the oxidation state of the central atom nickel is x. It is an anionic complex with two negative charges.
$x + 4( - 1) = - 2$
$x = + 2$.
Nickel is an element in the periodic table with atomic number \[28\] and electronic configuration \[\left[ {Ar} \right]3{d^8}4{s^2}\] . The electronic configuration of \[N{i^{2 + }}\] is\[\left[ {Ar} \right]3{d^8}\] . Thus the four cyanide atoms occupy the one \[d\], one \[s\] and two \[p\]-orbitals of the \[N{i^{2 + }}\] ion.

The \[{d_{{x^2} - {y^2}}}\] orbital is placed in the xy plane with the lobes directed along the x and y axes. Thus the \[ds{p^2}\] hybridization favors a planar geometry where all the four hybridized orbitals lie along the xy plane. Thus \[{d_{{x^2} - {y^2}}}\] orbital is used for \[ds{p^2}\] hybridization and the statement is correct.
(iii) Solid \[{N_2}{O_5}\] contains discrete units of \[N{O_2}^ + \] and \[N{O_3}^ - \].
\[{N_2}{O_5}\] is known as Dinitrogen pentoxide. The compound is a binary nitrogen oxide, which consists of nitrogen and oxygen. It exists as colourless crystals with melting point of \[41\;^\circ C\] and boiling point of \[47\;^\circ C\].
\[{N_2}{O_5}\] exists in the form of two structures depending on the phases of the compound. In the solid phase it is a salt called nitronium nitrate. It consists of separate nitronium cations \[{\left[ {N{O_2}} \right]^ + }\] and nitrate anions \[{\left[ {N{O_3}} \right]^ - }\] . The compound in the gas phase exists as a covalently bound molecule.
So, the correct answer is Option C.
Note: The concept of hybridization is important for determining the structure of the complex. The choice of \[d - \] orbitals depends on the type of hybridization and the geometry of the molecule.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




