
Why is carboxylic acid weaker than HCl?
Answer
527.7k+ views
Hint: The acidity or basicity of a chemical is going to depend on the capability of donation of ${{H}^{+}}$ and $O{{H}^{-}}$ respectively. If the chemical is going to donate the ${{H}^{+}}$ ions very easily then the chemical is called strong acid and vice versa.
Complete answer:
- In the question it is asked why carboxylic acid is weaker than HCl.
- First, we should know about the dissociation of the acid in water.
- The dissociation of the carboxylic acid in water is as follows.
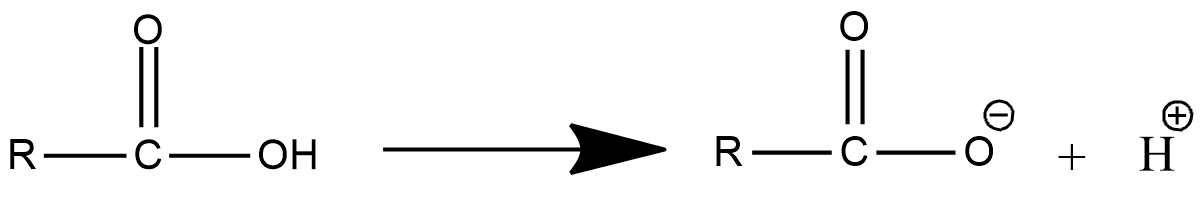
- In the above chemical dissociation of carboxylic acid, we can see that the carboxylic acid is going to convert into carboxylate anion and ${{H}^{+}}$ ion.
- But the carboxylate anion is going to exist in the following resonance structures.

- Due to the existence of the above chemical structures the ${{H}^{+}}$ ion formed at the time of dissociation is going to be grabbed by the formed resonance structures of the carboxylate anion.
- Coming to the dissociation of the HCl and it is as follows.
\[HCl\to {{H}^{+}}+C{{l}^{-}}\]
- The formed ${{H}^{+}}$ in the above chemical reaction is not going to be grabbed by the chloride ion which is formed in the above dissociation chemical reaction.
- Therefore, the carboxylate functional group is going to grab the ${{H}^{+}}$ ion which is formed after its dissociation but the HCl is not going to do like this.
- Therefore, the carboxylic acid is a weak acid when compared to HCl.
Note:
The strength of an acid is going to depend on the capability of the donation of the ${{H}^{+}}$ and not going to depend on the capability to accept the ${{H}^{+}}$ which is formed by itself during the dissociation in water.
Complete answer:
- In the question it is asked why carboxylic acid is weaker than HCl.
- First, we should know about the dissociation of the acid in water.
- The dissociation of the carboxylic acid in water is as follows.

- In the above chemical dissociation of carboxylic acid, we can see that the carboxylic acid is going to convert into carboxylate anion and ${{H}^{+}}$ ion.
- But the carboxylate anion is going to exist in the following resonance structures.

- Due to the existence of the above chemical structures the ${{H}^{+}}$ ion formed at the time of dissociation is going to be grabbed by the formed resonance structures of the carboxylate anion.
- Coming to the dissociation of the HCl and it is as follows.
\[HCl\to {{H}^{+}}+C{{l}^{-}}\]
- The formed ${{H}^{+}}$ in the above chemical reaction is not going to be grabbed by the chloride ion which is formed in the above dissociation chemical reaction.
- Therefore, the carboxylate functional group is going to grab the ${{H}^{+}}$ ion which is formed after its dissociation but the HCl is not going to do like this.
- Therefore, the carboxylic acid is a weak acid when compared to HCl.
Note:
The strength of an acid is going to depend on the capability of the donation of the ${{H}^{+}}$ and not going to depend on the capability to accept the ${{H}^{+}}$ which is formed by itself during the dissociation in water.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

The largest wind power cluster is located in the state class 11 social science CBSE

Explain zero factorial class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

What steps did the French revolutionaries take to create class 11 social science CBSE

Which among the following are examples of coming together class 11 social science CBSE




