Calculate total number of optical isomers in the following compounds:
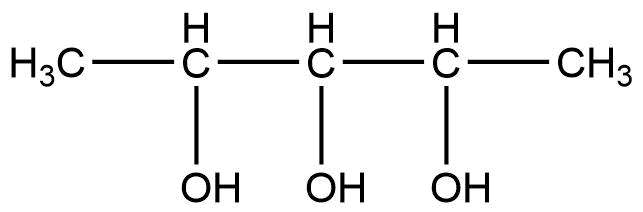
A \[{\text{2}}\]
B ${\text{4}}$
C ${\text{3}}$
D ${\text{8}}$

Answer
602.7k+ views
Hint: So here in this question we have to calculate the total number of optical isomers. Basically, optical isomers are two compounds which contain the same number and kinds of atoms, and bonds which have non – superimposable mirror images.
Complete step by step answer:
So in this compound there are 4 optical isomers and these are calculated when we multiply the number of chiral centers by two.
Mathematically, to calculate the total no. of optical isomers is ${{\text{2}}^{{\text{n - 1}}}}$, where n is equal to the number of chiral centers. So as in this compound there are three chiral carbons. So here we consider, ${\text{n = 3}}$
So by substituting the value of n, we get,
${\text{ = }}{{\text{2}}^{{\text{3 - 1}}}}$
${\text{ = }}{{\text{2}}^{\text{2}}}$
${\text{ = 4}}$
Additional Information: For optical isomers, molecules that are having chiral centers:-
If n is even, then the number of enantiomers ${\text{ = }}{{\text{2}}^{{\text{n - 1}}}}$
No. of meso compounds ${\text{ = }}{{\text{2}}^{{\text{n}} / {\text{2 - 1}}}}$
Total no. of optical isomers ${\text{ = }}{{\text{2}}^{{\text{n - 1}}}}{\text{ + }}{{\text{2}}^{{\text{n}} / {\text{2 - 1}}}}$
That is; number of enantiomers + number of meso compounds =Total no. of optical isomers.
If n is odd, then the number of enantiomers ${\text{ = }}{{\text{2}}^{{\text{n - 1}}}}{\text{ - }}{{\text{2}}^{\left( {{\text{n - 1}}} \right)}}^{ / {\text{2}}}$
No. of meso compounds ${\text{ = }}{{\text{2}}^{\left( {{\text{n - 1}}} \right) / {\text{2}}}}$
Total no. of optical isomers ${\text{ = }}{{\text{2}}^{{\text{n - 1}}}}$
So here, meso compounds are non-optically active members at least two of which are optically active.
And there are 2n optical isomers for an organic molecule with n chiral centers.
So, the correct answer is Option B .
Note:
Working on carbons is chiral, so we just have to check each one separately. A chiral carbon is not a branched group. A chiral carbon is a carbon atom that is bonded to four different atoms.
Complete step by step answer:
So in this compound there are 4 optical isomers and these are calculated when we multiply the number of chiral centers by two.
Mathematically, to calculate the total no. of optical isomers is ${{\text{2}}^{{\text{n - 1}}}}$, where n is equal to the number of chiral centers. So as in this compound there are three chiral carbons. So here we consider, ${\text{n = 3}}$
So by substituting the value of n, we get,
${\text{ = }}{{\text{2}}^{{\text{3 - 1}}}}$
${\text{ = }}{{\text{2}}^{\text{2}}}$
${\text{ = 4}}$
Additional Information: For optical isomers, molecules that are having chiral centers:-
If n is even, then the number of enantiomers ${\text{ = }}{{\text{2}}^{{\text{n - 1}}}}$
No. of meso compounds ${\text{ = }}{{\text{2}}^{{\text{n}} / {\text{2 - 1}}}}$
Total no. of optical isomers ${\text{ = }}{{\text{2}}^{{\text{n - 1}}}}{\text{ + }}{{\text{2}}^{{\text{n}} / {\text{2 - 1}}}}$
That is; number of enantiomers + number of meso compounds =Total no. of optical isomers.
If n is odd, then the number of enantiomers ${\text{ = }}{{\text{2}}^{{\text{n - 1}}}}{\text{ - }}{{\text{2}}^{\left( {{\text{n - 1}}} \right)}}^{ / {\text{2}}}$
No. of meso compounds ${\text{ = }}{{\text{2}}^{\left( {{\text{n - 1}}} \right) / {\text{2}}}}$
Total no. of optical isomers ${\text{ = }}{{\text{2}}^{{\text{n - 1}}}}$
So here, meso compounds are non-optically active members at least two of which are optically active.
And there are 2n optical isomers for an organic molecule with n chiral centers.
So, the correct answer is Option B .
Note:
Working on carbons is chiral, so we just have to check each one separately. A chiral carbon is not a branched group. A chiral carbon is a carbon atom that is bonded to four different atoms.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




