
How many bonding pairs and lone pairs surround the central atom in the $\text{I}_{3}^{-}$ ion?
A. 2, 2
B. 2, 3
C. 3, 2
D. 4, 3
Answer
603.9k+ views
Hint: Hybridisation is the mixing of an atomic orbital to form the chemical bonds according to the valence band theory. It forms a new orbital with the different geometry and shape of the hybridised molecule.
Complete answer:
-Firstly, we will calculate the hybridisation by using the formula that is
$\text{H = }\dfrac{1}{2}(\text{V + M +C - A)}$
-Here, H is the hybridisation, V is the valence electron of the central atom, M is the monovalent atom that is linked to the central atom, C is the charge present on the cation and A is the charge present on the anion.
-Now, we know that there are a total of 7 valence electrons in the iodine and it is a monovalent atom also.
-So, the value of V and M for iodine will be 7 and 2 respectively because two 2 molecules of iodine are linked to the central molecule of iodine.
-The value of C will be 1 because there is only a -1 charge on the iodine molecule.
-So, the value of H is:
$\text{H = }\dfrac{1}{2}(7\text{ }+\text{ }2\text{ + 1)}$
$\text{H = }\dfrac{1}{2}\text{ }\cdot \text{ 10 = 5}$
-The hybridisation of the centre iodine will be $\text{s}{{\text{p}}^{3}}\text{d}$ and it will have 3 lone pairs and 2 bond pairs with a linear shape.
-Central iodine makes 2 bonds and the 3 pairs remain unused.
-So, they arrange themselves such that there is minimum repulsion between the lone pairs
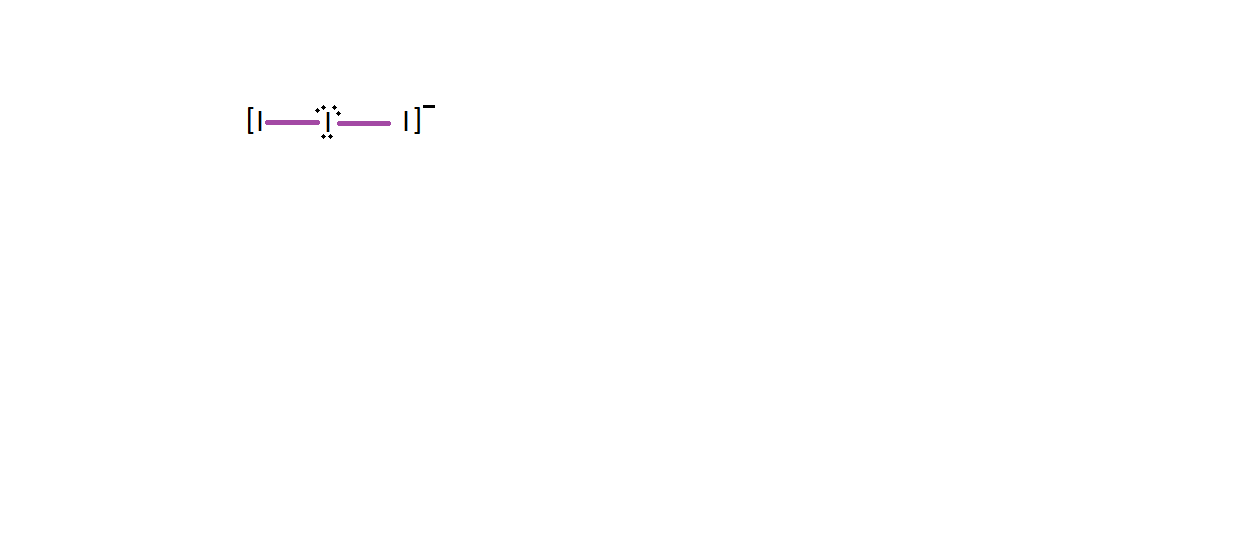
So, the correct answer is “Option B”.
Note: VSEPR or Valence Shell Electron Pair Theory tells us about the geometry of a molecule by arranging the molecules in such a way that there will be less repulsion between them.
Complete answer:
-Firstly, we will calculate the hybridisation by using the formula that is
$\text{H = }\dfrac{1}{2}(\text{V + M +C - A)}$
-Here, H is the hybridisation, V is the valence electron of the central atom, M is the monovalent atom that is linked to the central atom, C is the charge present on the cation and A is the charge present on the anion.
-Now, we know that there are a total of 7 valence electrons in the iodine and it is a monovalent atom also.
-So, the value of V and M for iodine will be 7 and 2 respectively because two 2 molecules of iodine are linked to the central molecule of iodine.
-The value of C will be 1 because there is only a -1 charge on the iodine molecule.
-So, the value of H is:
$\text{H = }\dfrac{1}{2}(7\text{ }+\text{ }2\text{ + 1)}$
$\text{H = }\dfrac{1}{2}\text{ }\cdot \text{ 10 = 5}$
-The hybridisation of the centre iodine will be $\text{s}{{\text{p}}^{3}}\text{d}$ and it will have 3 lone pairs and 2 bond pairs with a linear shape.
-Central iodine makes 2 bonds and the 3 pairs remain unused.
-So, they arrange themselves such that there is minimum repulsion between the lone pairs
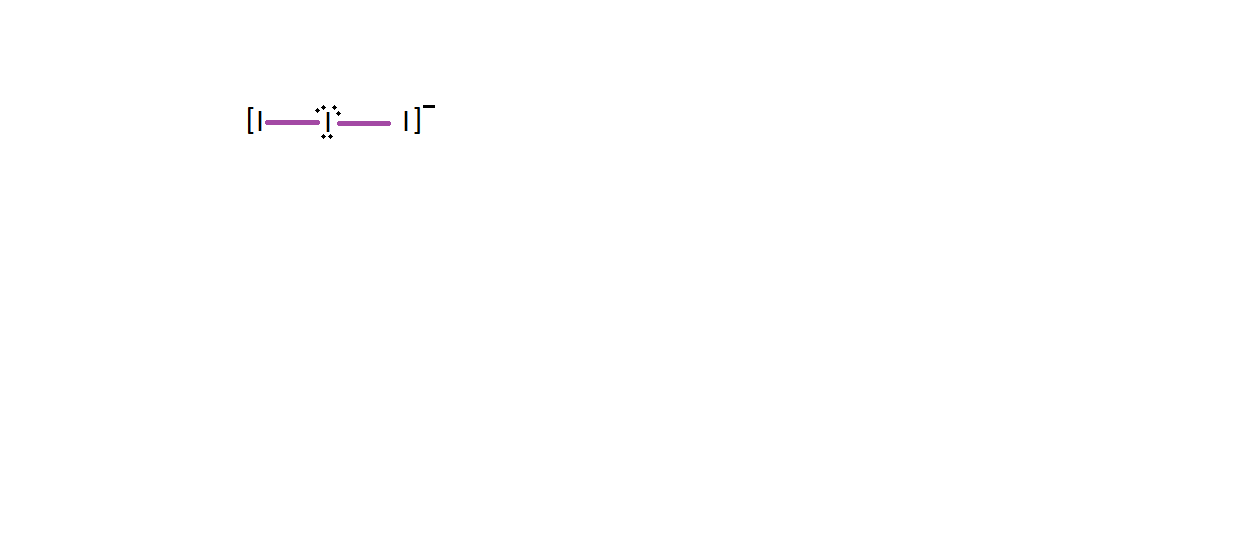
So, the correct answer is “Option B”.
Note: VSEPR or Valence Shell Electron Pair Theory tells us about the geometry of a molecule by arranging the molecules in such a way that there will be less repulsion between them.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

The largest wind power cluster is located in the state class 11 social science CBSE

Explain zero factorial class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

What steps did the French revolutionaries take to create class 11 social science CBSE

Which among the following are examples of coming together class 11 social science CBSE




