
A gas when passes through ${{K}_{2}}C{{r}_{2}}{{O}_{7}}$ and dil. ${{H}_{2}}S{{O}_{4}}$ solution turns it green, the gas is:
A.$C{{O}_{2}}$
B.$N{{H}_{3}}$
C.$S{{O}_{2}}$
D.$C{{l}_{2}}$
Answer
581.4k+ views
Hint: Potassium dichromate has an excellent property of oxidising other compounds. Chromium is present in the+4 oxidation state, where it itself gets reduced and the oxidation state changes to +3.
Complete answer:
In order to answer our question, we need to learn about potassium dichromate. The compound is prepared from chromite ore $(FeO.C{{r}_{2}}{{O}_{3}})$. Sodium dichromate is filtered and then treated with dil. ${{H}_{2}}S{{O}_{4}}$. Being less soluble, sodium sulphate separates out. Then hot concentrated solution is cooled when red crystals of sodium dichromate separates out on standing and then a hot concentrated solution of sodium dichromate is treated with potassium chloride to get potassium dichromate.
\[N{{a}_{2}}C{{r}_{2}}{{O}_{7}}+2KCl\to {{K}_{2}}C{{r}_{2}}{{O}_{7}}+2NaCl\]
The properties of potassium dichromate are:
i. It is an orange crystalline solid with melting point 670 K.
ii. It is appreciably soluble in hot water but moderately soluble in cold water.
iii. On heating K, Cr,o, decomposes to give potassium chromate and chromic oxide.
\[4{{K}_{2}}C{{r}_{2}}{{O}_{7}}+\Delta \to 4{{K}_{2}}C{{r}_{2}}{{O}_{4}}+2C{{r}_{2}}{{O}_{3}}+3{{O}_{2}}\]
iv. Potassium dichromate acts as a strong oxidising agent in acidic medium.
v. When a mixture of metal chloride is heated with potassium dichromate and concentrated sulphuric acid, the orange red fumes of chromyl chloride are formed.
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}+4NaCl+6{{H}_{2}}S{{O}_{4}}\to 2KHS{{O}_{4}}+4NaHS{{O}_{4}}+2Cr{{O}_{2}}C{{l}_{2}}+3{{H}_{2}}O\]
The structure of potassium dichromate is:
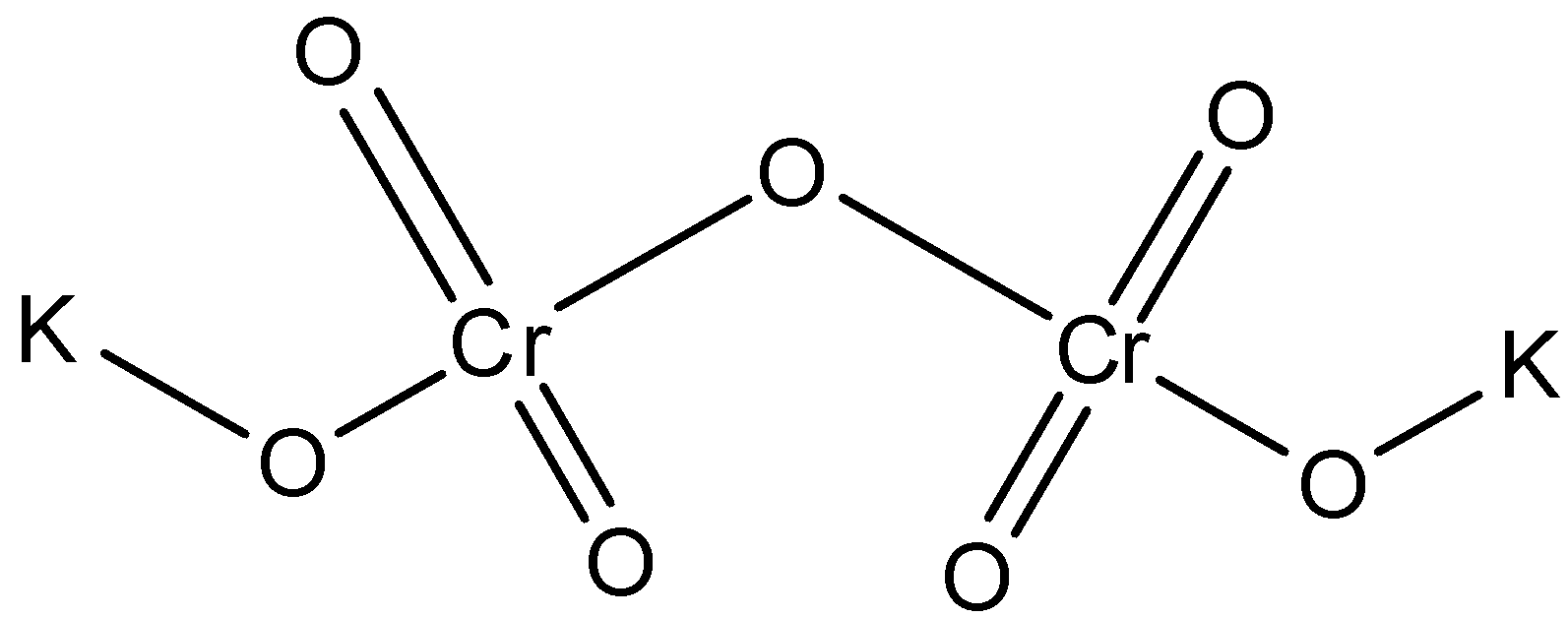
Now, let us come to our question. In the compound, chromium is present in the +4 oxidation state. However, when sulphur dioxide gas is passed through it, then the oxidation state of chromium changes to +3 and the colour of the solution changes to green. The reaction is represented as:
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}+2{{H}_{2}}S{{O}_{4}}+3S{{O}_{2}}\to 2C{{r}_{2}}{{(S{{O}_{4}})}_{3}}+{{K}_{2}}S{{O}_{4}}+{{H}_{2}}O\]
So, we get the correct answer as option C.
NOTE: The uses of potassium dichromate are:
i.For the volumetric estimation of ferrous salts, halides and sulphides.
ii. For the preparation of other chromium compounds such as chrome alum chrome yellow etc
iii. Used in chrome tanning in leather industry
iv. Used as an oxidising agent.
Complete answer:
In order to answer our question, we need to learn about potassium dichromate. The compound is prepared from chromite ore $(FeO.C{{r}_{2}}{{O}_{3}})$. Sodium dichromate is filtered and then treated with dil. ${{H}_{2}}S{{O}_{4}}$. Being less soluble, sodium sulphate separates out. Then hot concentrated solution is cooled when red crystals of sodium dichromate separates out on standing and then a hot concentrated solution of sodium dichromate is treated with potassium chloride to get potassium dichromate.
\[N{{a}_{2}}C{{r}_{2}}{{O}_{7}}+2KCl\to {{K}_{2}}C{{r}_{2}}{{O}_{7}}+2NaCl\]
The properties of potassium dichromate are:
i. It is an orange crystalline solid with melting point 670 K.
ii. It is appreciably soluble in hot water but moderately soluble in cold water.
iii. On heating K, Cr,o, decomposes to give potassium chromate and chromic oxide.
\[4{{K}_{2}}C{{r}_{2}}{{O}_{7}}+\Delta \to 4{{K}_{2}}C{{r}_{2}}{{O}_{4}}+2C{{r}_{2}}{{O}_{3}}+3{{O}_{2}}\]
iv. Potassium dichromate acts as a strong oxidising agent in acidic medium.
v. When a mixture of metal chloride is heated with potassium dichromate and concentrated sulphuric acid, the orange red fumes of chromyl chloride are formed.
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}+4NaCl+6{{H}_{2}}S{{O}_{4}}\to 2KHS{{O}_{4}}+4NaHS{{O}_{4}}+2Cr{{O}_{2}}C{{l}_{2}}+3{{H}_{2}}O\]
The structure of potassium dichromate is:

Now, let us come to our question. In the compound, chromium is present in the +4 oxidation state. However, when sulphur dioxide gas is passed through it, then the oxidation state of chromium changes to +3 and the colour of the solution changes to green. The reaction is represented as:
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}+2{{H}_{2}}S{{O}_{4}}+3S{{O}_{2}}\to 2C{{r}_{2}}{{(S{{O}_{4}})}_{3}}+{{K}_{2}}S{{O}_{4}}+{{H}_{2}}O\]
So, we get the correct answer as option C.
NOTE: The uses of potassium dichromate are:
i.For the volumetric estimation of ferrous salts, halides and sulphides.
ii. For the preparation of other chromium compounds such as chrome alum chrome yellow etc
iii. Used in chrome tanning in leather industry
iv. Used as an oxidising agent.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Draw a diagram of nephron and explain its structur class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

Chemical formula of Bleaching powder is A Ca2OCl2 B class 11 chemistry CBSE

Name the part of the brain responsible for the precision class 11 biology CBSE

The growth of tendril in pea plants is due to AEffect class 11 biology CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE




