What is A?
$C{{H}_{3}}CHO+C{{H}_{3}}MgBr\to \text{ Product }\xrightarrow{{{H}_{2}}O}A$
(a)- Primary alcohol
(b)- Secondary alcohol
(c)- Tertiary alcohol
(d)- Ketone
Answer
560.1k+ views
Hint: The given compound in the reaction is ethanal and ethanal is a primary compound. $C{{H}_{3}}MgBr$ is a Grignard reagent, when this reacts with carbonyl group then there will be the addition of alkyl group at the carbon atom having the oxygen atom.
Complete answer: In the reaction given in the question, the reactant is ethanal because there are two carbon atoms and there is an aldehyde functional group, and this ethanal reacts with $C{{H}_{3}}MgBr$ which is a Grignard reagent. The Grignard reagent is a compound in which an alkyl group is present with magnesium bromide. The alkyl group present with MgBr in this compound is methyl.
When this reacts with the carbonyl group then there will be the addition of an alkyl group at the carbon atom having the oxygen atom. Now, when the ethanal reacts with methyl magnesium bromide, then the methyl group will attack the carbon atom having the carbonyl group, while the MgBr will attack the Oxygen atom.
When this molecule is treated with water then the –OMgBr group will be hydrolyzed and it will convert into the –OH group. The series of reaction is given below:
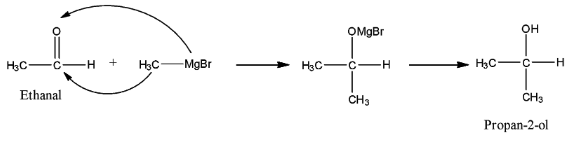
So, the product formed is propan-2-ol, therefore, the hydroxyl group is attached with a carbon atom that is further attached with two carbon atoms. This means that the alcohol formed is secondary alcohol.
Therefore, the correct answer is an option (b).
Note: To form primary alcohol by using Grignard reagent then we have to use methanol as reactant because the only methanol can form primary alcohol, and with ketones, always tertiary alcohols are formed.
Complete answer: In the reaction given in the question, the reactant is ethanal because there are two carbon atoms and there is an aldehyde functional group, and this ethanal reacts with $C{{H}_{3}}MgBr$ which is a Grignard reagent. The Grignard reagent is a compound in which an alkyl group is present with magnesium bromide. The alkyl group present with MgBr in this compound is methyl.
When this reacts with the carbonyl group then there will be the addition of an alkyl group at the carbon atom having the oxygen atom. Now, when the ethanal reacts with methyl magnesium bromide, then the methyl group will attack the carbon atom having the carbonyl group, while the MgBr will attack the Oxygen atom.
When this molecule is treated with water then the –OMgBr group will be hydrolyzed and it will convert into the –OH group. The series of reaction is given below:

So, the product formed is propan-2-ol, therefore, the hydroxyl group is attached with a carbon atom that is further attached with two carbon atoms. This means that the alcohol formed is secondary alcohol.
Therefore, the correct answer is an option (b).
Note: To form primary alcohol by using Grignard reagent then we have to use methanol as reactant because the only methanol can form primary alcohol, and with ketones, always tertiary alcohols are formed.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




