How many 3C - 4${{\text{e}}^{-}}$ bonds are present in the dimer of $\text{BeC}{{\text{l}}_{2}}$?
Answer
616.8k+ views
Hint: 3C-4${{\text{e}}^{-}}$ bonds stands for three centre - 4 electron bonds. Coordination bonds are those bonds which are made after the sharing of lone pair electrons. 3C-4${{\text{e}}^{-}}$ bond means that in between 3 atoms total 4 electrons are shared which do not participate in the reaction.
Complete answer:
-Firstly, we have to draw the structure of beryllium chloride.
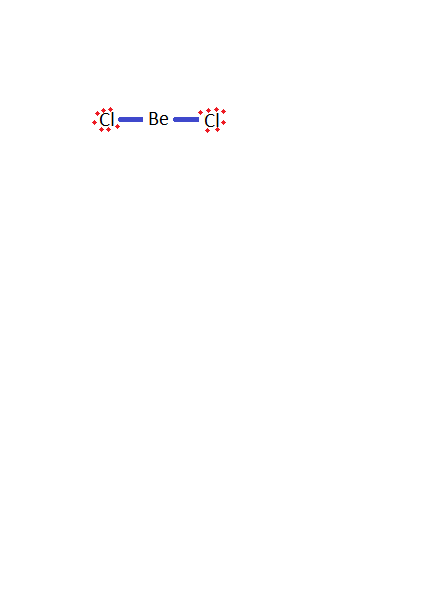
-Now, here chlorine has a lone pair of an electron which can be shared with the other molecules which are electron deficient.
-So, beryllium chloride dimerises to form \[\text{B}{{\text{e}}_{2}}\text{C}{{\text{l}}_{4}}\] because it is unstable due to incomplete octet present in the boron and the structure of it is:
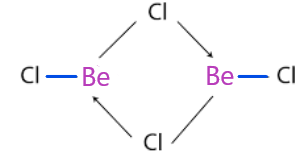
-So, we can see that the two molecules of chlorine share the lone pair of an electron with boron to fulfill the octet.
-Also, the compound is an electron-deficient molecule because electron-deficient molecules are those in which the total no. of atoms in the boron is less than 8.
-As we can also see that four-electrons are being shared, that is two electrons from each chlorine atom and they are given to the boron.
-So, a total of 3 atoms participated that's why it is called 3 centre - 4 electron bond.
-And a total of two bonds are formed.
Therefore, total 2, 3C - 4${{\text{e}}^{-}}$ bonds in the structure of beryllium chloride.
Note: Electron deficient molecules tend to accept electrons so they can make coordinate bonds easily. Other molecules which can form 3C - 4${{\text{e}}^{-}}$bonds are \[\text{Xe}{{\text{F}}_{2}}\text{, A}{{\text{l}}_{2}}\text{C}{{\text{l}}_{6}}\text{, HF, etc}\]. Electron rich molecules are those species which have an extra pair of electrons to make the bond with electron deficient molecules.
Complete answer:
-Firstly, we have to draw the structure of beryllium chloride.

-Now, here chlorine has a lone pair of an electron which can be shared with the other molecules which are electron deficient.
-So, beryllium chloride dimerises to form \[\text{B}{{\text{e}}_{2}}\text{C}{{\text{l}}_{4}}\] because it is unstable due to incomplete octet present in the boron and the structure of it is:

-So, we can see that the two molecules of chlorine share the lone pair of an electron with boron to fulfill the octet.
-Also, the compound is an electron-deficient molecule because electron-deficient molecules are those in which the total no. of atoms in the boron is less than 8.
-As we can also see that four-electrons are being shared, that is two electrons from each chlorine atom and they are given to the boron.
-So, a total of 3 atoms participated that's why it is called 3 centre - 4 electron bond.
-And a total of two bonds are formed.
Therefore, total 2, 3C - 4${{\text{e}}^{-}}$ bonds in the structure of beryllium chloride.
Note: Electron deficient molecules tend to accept electrons so they can make coordinate bonds easily. Other molecules which can form 3C - 4${{\text{e}}^{-}}$bonds are \[\text{Xe}{{\text{F}}_{2}}\text{, A}{{\text{l}}_{2}}\text{C}{{\text{l}}_{6}}\text{, HF, etc}\]. Electron rich molecules are those species which have an extra pair of electrons to make the bond with electron deficient molecules.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




