\[3 - {\rm{methylpent}} - 2 - {\rm{ene}}\] on reaction with ${\rm{HBr}}$ in presence of peroxide forms an addition product. The number of possible stereoisomers for the products:
(A) Zero
(B) Two
(C) Four
(D) Six
Answer
585.9k+ views
Hint:We know that in case peroxide is present, addition of ${\rm{HBr}}$ follows anti-Markovnikov rule and number of possible stereoisomers can be deduced from the number of chiral centres.
Complete step-by-step answer:Let’s start by having a look at our given alkene that is \[3 - {\rm{methylpent}} - 2 - {\rm{ene}}\]. We can draw the structure for this alkene as follows:
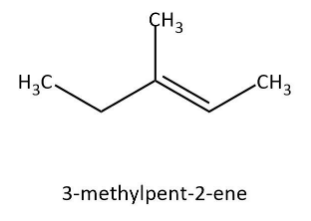
As we can see that it is an unsymmetrical alkene, so we have to be careful with the addition of ${\rm{HBr}}$ to this. Usually we would follow Markovnikov rule but now we are doing addition in the presence of peroxide. So, we have to follow the anti-Markovnikov rule and we will see that less substituted carbon will get priority. Let’s write the addition reaction as follows:
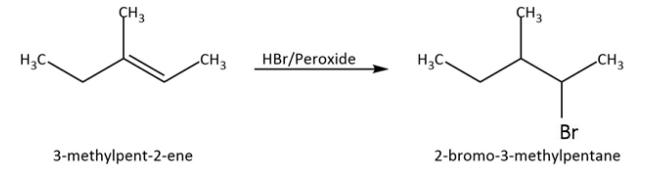
Now that we have our addition product, let’s find out how many chiral centres are there in this molecule by identifying carbon atoms that are bonded to four different substituents.
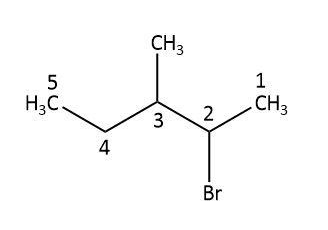
In the addition product, we can see that the first carbon has three hydrogen atoms, second and third carbons have four different substituents, fourth has two hydrogen atoms and fifth has three hydrogen atoms. So, we can say that we have two chiral centres in our addition product.
Now we can find out the number of possible stereoisomers as ${2^2} = 4$, to be four. We can draw the structures of the four stereoisomers as follows:
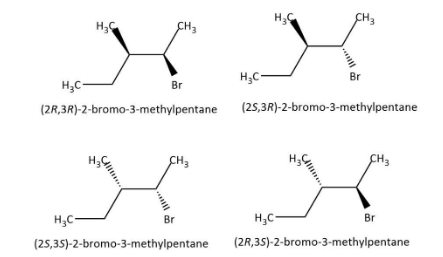
Hence, the correct option is C.
Note: We can understand the formation of the product according to the anti-Markovnikov rule as participation of peroxide in the free radical mechanism and giving rise to peroxide effect.
Complete step-by-step answer:Let’s start by having a look at our given alkene that is \[3 - {\rm{methylpent}} - 2 - {\rm{ene}}\]. We can draw the structure for this alkene as follows:

As we can see that it is an unsymmetrical alkene, so we have to be careful with the addition of ${\rm{HBr}}$ to this. Usually we would follow Markovnikov rule but now we are doing addition in the presence of peroxide. So, we have to follow the anti-Markovnikov rule and we will see that less substituted carbon will get priority. Let’s write the addition reaction as follows:

Now that we have our addition product, let’s find out how many chiral centres are there in this molecule by identifying carbon atoms that are bonded to four different substituents.

In the addition product, we can see that the first carbon has three hydrogen atoms, second and third carbons have four different substituents, fourth has two hydrogen atoms and fifth has three hydrogen atoms. So, we can say that we have two chiral centres in our addition product.
Now we can find out the number of possible stereoisomers as ${2^2} = 4$, to be four. We can draw the structures of the four stereoisomers as follows:

Hence, the correct option is C.
Note: We can understand the formation of the product according to the anti-Markovnikov rule as participation of peroxide in the free radical mechanism and giving rise to peroxide effect.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
State and prove Bernoullis theorem class 11 physics CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Which of the following is the hottest month AMay BJune class 11 social science CBSE

What is 1s 2s 2p 3s 3p class 11 chemistry CBSE

Draw the molecular orbital diagram of N2N2 + N2 Write class 11 chemistry CBSE

Which gland is known as mixed gland class 11 biology CBSE




