



Key Concepts and Examples of Post Transition Metals for Students
A group of elements in the periodic table are known as the post-transition metals. They are situated to the left of the metalloids and the right of the transition metals. In addition, they are referred to as "other" and "poor" metals. The name for this class of metals (poor, other, post-transition) and which elements should be included are up for debate among scientists.
Typically, any metal from groups 13, 14, and 15—aluminium, gallium, indium, tin, thallium, lead, and bismuth—constitutes post-transition metals. Additionally, polonium is frequently categorised as a post-transition metal. The representation of post-transition metals is shown below.
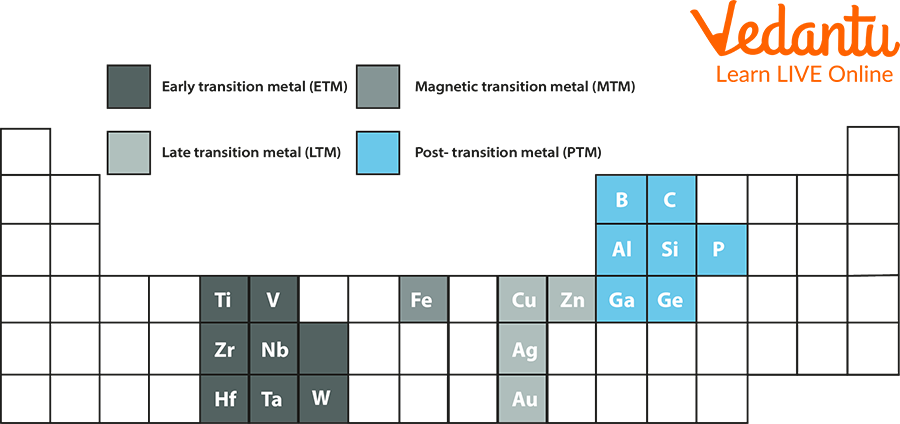
Post-transition Metals
Similar Properties of Post Transition Metal
Post-transition metals have a lot of similar qualities, like:
Under normal circumstances, they appear as solid metal.
They are ductile, malleable, and effective heat and electrical conductors, like most metals.
They're fairly densely packed together.
They typically are softer and have lower melting and boiling temperatures than transition metals.
Periodic Trends of Post Transition Metals
Since there are typically fewer electrons available for metallic bonding when atomic radii decrease and ionisation energy increase, ions are typically smaller, more polarising, and more likely to form covalent connections. As a result, they exhibit less metallic nature.
Order of Abundance
Aluminium, which is also the most common metal in the Earth's crust, is the most common of the post-transition metals on Earth. The abundance of post-transition metals in the Earth's crust is listed below in order:
Aluminium
Gallium
Lead
Tin
Thallium
Indium
Bismuth
Uses of Post-Transition Metals
There are numerous uses of the various elements of this family. For creating kitchenware, electronics, soldering, and plating steel, aluminium and tin are employed. Pepto-Bismol, a medication used to settle upset stomachs, is made from bismuth. Gallium is utilised in semiconductors and fuel cells, while Indium is used in electronics, such as touch screens and flat panel displays. Among other things, lead is utilised in the production of batteries.
Transition Elements List
Interesting Facts About Post-Transition Metals
Now, let’s read some interesting facts about post-transition metals
Sometimes post-transition metals are used to group zinc, cadmium, and mercury instead of transition metals.
After silicon and oxygen, aluminium is the third most common element in the crust of the Earth.
In some cases, metalloids rather than post-transition metals are used to describe germanium and antimony.
The melting point of gallium is just slightly over room temperature, and it will melt in your hand.
Pepto-Bismol, a medication used to soothe upset stomachs, contains bismuth.
Formerly believed to be the heaviest stable element, bismuth has lately been shown to have a small amount of radioactivity.
Electronics such as touch screens and flat panel displays employ indium.
The Greek word "thallos," which means "a green sprout or twig," is where the name "thallium" originates.
Thallium is extremely poisonous and lethal. Hair loss is one sign of thallium toxicity.
Summary
In this article we have discussed similar example of transition metal, transition elements list, order of abundance, uses and interesting facts about post transition metal. The set of metals on the periodic table is referred to as post-transition metals or poor metals. The transition metals are to their right of them. We also learned what is meant by transition elements. We learned that when compared to transition metals, post-transition metals often have higher electronegativities.
Furthermore, their melting points are lower. In general, they are softer than other metals. Lead, tin, and other soft metals are among them.
FAQs on Interesting Facts About Post Transition Metals
1. What are post-transition metals?
Post-transition metals are a group of chemical elements located in the p-block of the periodic table. They are positioned to the right of the transition metals and to the left of the metalloids. These metals are distinct because they have a completely filled d-electron subshell, which influences their chemical properties and differentiates them from true transition metals.
2. Which elements are considered post-transition metals and where are they found on the periodic table?
Post-transition metals are found in Groups 13, 14, and 15, occupying the space between the transition metals and metalloids. Key examples include:
- Group 13: Aluminium (Al), Gallium (Ga), Indium (In), Thallium (Tl)
- Group 14: Tin (Sn), Lead (Pb)
- Group 15: Bismuth (Bi)
3. What are the main characteristics that define post-transition metals?
Post-transition metals display properties that are often intermediate between typical metals and non-metals. Their primary characteristics are:
- They are generally softer and have lower melting and boiling points compared to the neighbouring transition metals.
- They are more electronegative than transition metals.
- They show a greater tendency to form covalent bonds.
- Their crystal structures are often more complex and less densely packed than those of most other metals.
4. What is the key difference between a transition metal and a post-transition metal?
The fundamental difference lies in their electron configuration. A transition metal is defined as an element that has an incompletely filled d-orbital in either its neutral atomic state or in any of its common oxidation states. In contrast, a post-transition metal has a completely filled d-subshell (d¹⁰). This structural difference leads to significant variations in properties like magnetism, compound colour, and catalytic ability.
5. Why are post-transition metals sometimes called 'poor metals'?
The term 'poor metals' is an informal name that highlights how their properties differ from more traditionally 'metallic' elements. They are called this because they generally have lower mechanical strength, are softer, and possess lower melting points compared to the more robust transition metals. Their higher electronegativity and tendency to form covalent bonds also distinguish them from the strong metallic bonding seen in other metal groups.
6. How does the 'inert pair effect' influence the chemistry of heavier post-transition metals?
The inert pair effect is a crucial concept for understanding heavier post-transition metals like Thallium (Tl), Lead (Pb), and Bismuth (Bi). It is the tendency of the two electrons in the outermost s-orbital to remain un-ionised or unshared in compounds. This happens because these s-electrons are more tightly held by the nucleus. Consequently, these elements exhibit stable oxidation states that are two less than their group valency, such as Tl⁺¹, Pb⁺², and Bi⁺³ being more common and stable than Tl⁺³, Pb⁺⁴, and Bi⁺⁵ respectively.
7. Is Zinc (Zn) a post-transition metal? Explain its classification.
The classification of Zinc is a common point of confusion. According to the strict IUPAC definition, Zinc is not a transition metal because it has a completely filled d-orbital (3d¹⁰) in its atomic state and its common +2 ion. While it shares this filled d-shell trait with post-transition metals, it is officially a Group 12 element that marks the end of the d-block. Therefore, while it is sometimes grouped with them for discussion, it is more accurately classified separately from the p-block post-transition metals like Aluminium or Tin.
8. What are some important real-world applications of post-transition metals?
Post-transition metals are essential in many industrial and consumer applications. Key examples include:
- Aluminium (Al): Used in aircraft, construction materials, and packaging (foil, cans) due to its low density and corrosion resistance.
- Tin (Sn): Used for coating steel to prevent rust (tin cans) and in alloys like solder for electronics.
- Lead (Pb): Primarily used today in lead-acid batteries for vehicles and as an effective shield against radiation.
- Gallium (Ga): Critical for modern electronics, used in semiconductors like gallium arsenide (GaAs) for LEDs and integrated circuits.









