Write the empirical formula for acetic acid.
Answer
636.6k+ views
Hint: Having a basic idea about the structure of Acetic Acid will give us an idea how to form the empirical formula. It is even advised to recall the different rules of nomenclature.
Complete step-by-step answer:
Here is the structure of Acetic Acid:
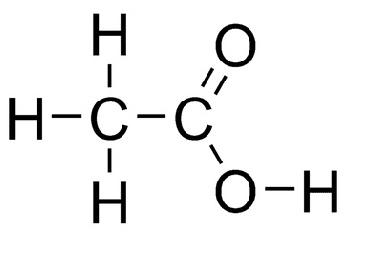
From the structure we can write the formula of Acetic Acid, which is:
${\text{C}}{{\text{H}}_{\text{3}}}{\text{COOH}}$
In chemistry the empirical formula of a chemical compound is the simplest positive integer ratio of atoms present in a compound.
There are 4 hydrogen atoms, 2 carbon atoms and 2 oxygen atoms.
So, we can write the empirical formula of Acetic Acid as:
${{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}{{\text{O}}_{\text{2}}}$
Note: So, here are some properties of acetic acid that we should know:
Acetic acid is important for microorganisms in food industry because of their ability to oxidize many types of sugars and alcohol to organic acids as end product during fermentation process
Acetic acid can be a hazardous chemical if not used in a safe and appropriate manner. This liquid is highly corrosive to the skin and eyes and because of this must be handled with extreme care acetic acid can also be damaging to the internal organs are interested or in the case of vapour inhalation.
Acetic acid is used in vinegar which is used as a condiment and in the pickling of raw vegetables and other foods. Acetic acid is used for the manufacture of inks and dyes.
We should also know that acetic acid is a weak monoprotic acid.
Complete step-by-step answer:
Here is the structure of Acetic Acid:

From the structure we can write the formula of Acetic Acid, which is:
${\text{C}}{{\text{H}}_{\text{3}}}{\text{COOH}}$
In chemistry the empirical formula of a chemical compound is the simplest positive integer ratio of atoms present in a compound.
There are 4 hydrogen atoms, 2 carbon atoms and 2 oxygen atoms.
So, we can write the empirical formula of Acetic Acid as:
${{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}{{\text{O}}_{\text{2}}}$
Note: So, here are some properties of acetic acid that we should know:
Acetic acid is important for microorganisms in food industry because of their ability to oxidize many types of sugars and alcohol to organic acids as end product during fermentation process
Acetic acid can be a hazardous chemical if not used in a safe and appropriate manner. This liquid is highly corrosive to the skin and eyes and because of this must be handled with extreme care acetic acid can also be damaging to the internal organs are interested or in the case of vapour inhalation.
Acetic acid is used in vinegar which is used as a condiment and in the pickling of raw vegetables and other foods. Acetic acid is used for the manufacture of inks and dyes.
We should also know that acetic acid is a weak monoprotic acid.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

DNA is not present in A Nucleus B Mitochondria C Chloroplast class 11 biology CBSE




