Write a note on Friedel Crafts acylation.
Answer
623.4k+ views
Hint: The two primary types of Friedel-Crafts reactions are the alkylation and acylation reactions. Friedel craft acylation is a type of an organic reaction in which an acyl group $( - COR)$is attached to a benzene ring.
Complete answer:
As we know, Friedel craft acylation is a type of an organic reaction in which an acyl group is attached to a benzene ring. This can be easily understood with the help of an example: When benzene is reacted with an acyl halide in the presence of anhydrous aluminium chloride, it gives acyl benzene as a product. And it is a type of electrophilic substitution reaction, electrophiles, as we know, are electron-loving. This can be referred to in other words as electron-deficient species and thus can accept an electron pair from electron-rich species. Some of the examples are carbocations and carbonyl compounds, and the term substitution refers to the removal of a hydrogen atom and is replaced by group or an atom in this case hydrogen is replaced by an acyl group. The reaction for the above-mentioned example is as follows:
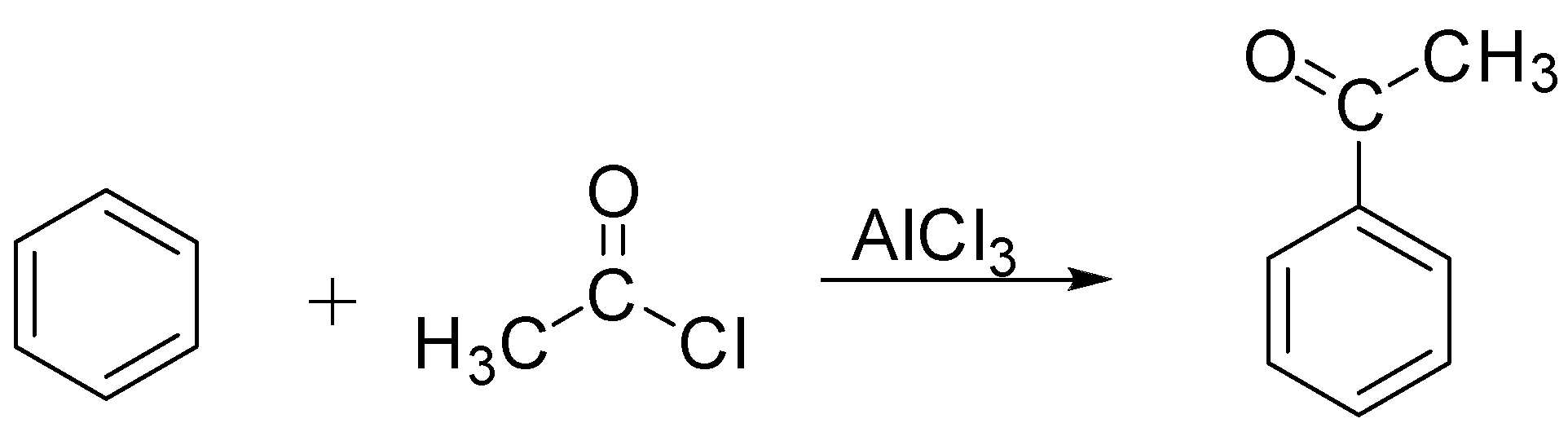
Here Acyl group is an electrophile as there is a formation of partial positive charge on the carbon atom of the acyl group. Acylation reactions can also be carried out with acetic anhydride as a substrate in the presence of Anhydrous $AlC{l_3}$ .
Note:
There are many types of Friedel Crafts reaction but a student can confuse between the alkylation and the acylation reaction these are quite a similar type of reaction as in Alkylation there is a substitution of hydrogen by an alkyl group and in acylation, there is a substitution of hydrogen by an acyl group.
Complete answer:
As we know, Friedel craft acylation is a type of an organic reaction in which an acyl group is attached to a benzene ring. This can be easily understood with the help of an example: When benzene is reacted with an acyl halide in the presence of anhydrous aluminium chloride, it gives acyl benzene as a product. And it is a type of electrophilic substitution reaction, electrophiles, as we know, are electron-loving. This can be referred to in other words as electron-deficient species and thus can accept an electron pair from electron-rich species. Some of the examples are carbocations and carbonyl compounds, and the term substitution refers to the removal of a hydrogen atom and is replaced by group or an atom in this case hydrogen is replaced by an acyl group. The reaction for the above-mentioned example is as follows:

Here Acyl group is an electrophile as there is a formation of partial positive charge on the carbon atom of the acyl group. Acylation reactions can also be carried out with acetic anhydride as a substrate in the presence of Anhydrous $AlC{l_3}$ .
Note:
There are many types of Friedel Crafts reaction but a student can confuse between the alkylation and the acylation reaction these are quite a similar type of reaction as in Alkylation there is a substitution of hydrogen by an alkyl group and in acylation, there is a substitution of hydrogen by an acyl group.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




