Which oxyacid of sulphur contains S-S single bond?
A. Oleum
B. Marshall’s acid
C. Dithionic acid
D. Thiosulphuric acid
Answer
622.2k+ views
Hint: Oxyacid is a compound that contains oxygen, hydrogen and one other element. Here, we are provided with oxoacids of sulphur, these contain oxygen, hydrogen and sulphur atoms.
Complete step by step answer:
- Oleum is also called as pyrosulfuric acid. The molecular formula of oleum is ${{H}_{2}}{{S}_{2}}{{O}_{7}}$. We can see the structure of oleum:
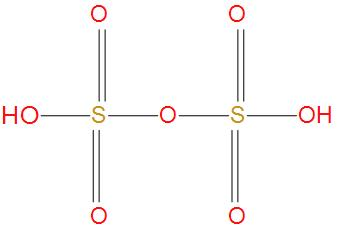
- We can see from the structure that there is no S-S single bond present, hence this option is wrong.
- Next option is Marshall’s acid, we also called it as peroxydisulfuric acid, its molecular formula is ${{H}_{2}}{{S}_{2}}{{O}_{8}}$ . We will see the structure of this:
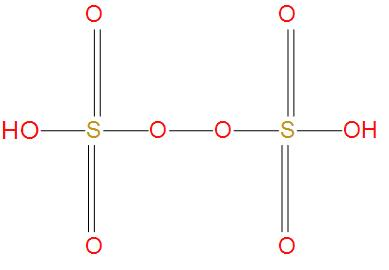
- Here, we can see that there is one peroxy linkage that O-O is present, and there is no S-S single bond present. Hence, this option is wrong.
- Let’s see the next option Dithionic acid, its molecular formula is ${{H}_{2}}{{S}_{2}}{{O}_{6}}$. We will see the structure of this:
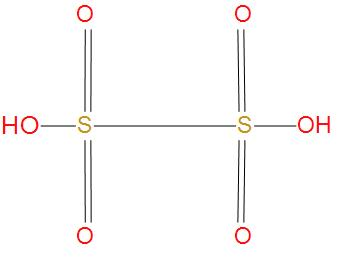
- We can see from the structure that there is one S-S single bond present. And we can say that this option is correct.
- Similarly, in Thiosulphuric acid, there is no S-S single bond present. We can see this from structure:
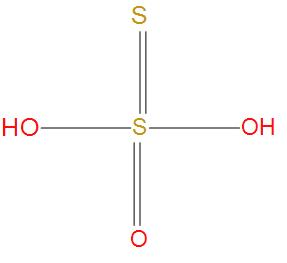
Hence, we can conclude that the correct option is (C), Dithionic acid contains S-S single bond.
Note: We know that an oxyacid molecule contains the structure X-O-H, where O is oxygen, H is hydrogen and X is any element. Where, other atoms can be connected to the central metal atom X. In a solution, oxyacid can be dissociated as: $X-O-H\rightleftarrows {{\left( X-O \right)}^{-}}+H$
Complete step by step answer:
- Oleum is also called as pyrosulfuric acid. The molecular formula of oleum is ${{H}_{2}}{{S}_{2}}{{O}_{7}}$. We can see the structure of oleum:

- We can see from the structure that there is no S-S single bond present, hence this option is wrong.
- Next option is Marshall’s acid, we also called it as peroxydisulfuric acid, its molecular formula is ${{H}_{2}}{{S}_{2}}{{O}_{8}}$ . We will see the structure of this:

- Here, we can see that there is one peroxy linkage that O-O is present, and there is no S-S single bond present. Hence, this option is wrong.
- Let’s see the next option Dithionic acid, its molecular formula is ${{H}_{2}}{{S}_{2}}{{O}_{6}}$. We will see the structure of this:

- We can see from the structure that there is one S-S single bond present. And we can say that this option is correct.
- Similarly, in Thiosulphuric acid, there is no S-S single bond present. We can see this from structure:

Hence, we can conclude that the correct option is (C), Dithionic acid contains S-S single bond.
Note: We know that an oxyacid molecule contains the structure X-O-H, where O is oxygen, H is hydrogen and X is any element. Where, other atoms can be connected to the central metal atom X. In a solution, oxyacid can be dissociated as: $X-O-H\rightleftarrows {{\left( X-O \right)}^{-}}+H$
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




