Which of the following ring compounds obeys Huckel’s rule?
(A)- ${{C}_{4}}H_{4}^{-1}$
(B)- ${{C}_{4}}H_{4}^{+1}$
(C)- ${{C}_{4}}H_{4}^{-2}$
(D)- ${{C}_{4}}{{H}_{4}}$
Answer
611.7k+ views
Hint: Erich Huckel in 1931 proposed a condition for a compound to be aromatic popularly known as Huckel’s rule. It states that a cyclic, planar and conjugated system having (4n+2)$\pi $ electrons, is considered to be aromatic.
Complete answer:
Aromatic compounds must obey Huckel’s rule and contain a certain number of $\pi $ electrons which should be equal to 4n+2. Here, n can be zero or any other positive integer. Therefore, compounds containing$2\pi $, $6\pi $, $10\pi $, $14\pi $ and so on electrons are aromatic.
Let us try to find the ring compound which obeys Huckel’s rule form the above given options.
${{C}_{4}}H_{4}^{-1}$
Possible ring structures with ${{C}_{4}}H_{4}^{-1}$ are shown below:
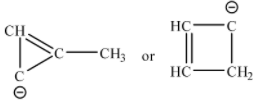
Neither of the compounds follow Huckel’s rule. In the three-membered ring compound the total number of $\pi $ electrons is 4, which is not a multiple of (4n+2)$\pi $. Ring compounds containing $4\pi $ electrons are anti-aromatic in nature. In the four-membered ring, there is a $s{{p}^{3}}$ hybridized tetrahedral carbon, so the ring is no longer planar. Thus, Huckel’s rule cannot be applied.
${{C}_{4}}H_{4}^{+1}$
Possible structure for ${{C}_{4}}H_{4}^{+1}$ is:
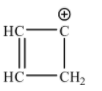
This ring is not planar as it contains a $s{{p}^{3}}$ hybridized carbon. Therefore, Huckel’s rule is not applicable.
${{C}_{4}}H_{4}^{-2}$
With the given molecular formula the possible ring structure is shown below:
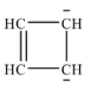
This ring compound contains $6\pi $ electrons which is equal to (4n+2)$\pi $ when n=1. Therefore, it obeys Huckel’s rule and is aromatic.
${{C}_{4}}{{H}_{4}}$
Structure of ${{C}_{4}}{{H}_{4}}$ is given as:
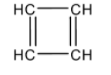
This cyclic compound contains $4\pi $ electrons and thus, it is antiaromatic. This compound also does not follow Huckel’s rule.
The only compound following Huckel’s rule is ${{C}_{4}}H_{4}^{-2}$.
Hence, the correct option is (C).
Additional information:
Compounds which lacks one or more of the four requirements, i.e. cyclic, planar, conjugated and (4n+2)$\pi $ electrons for aromatic; 4n$\pi $ for antiaromatic, to be aromatic or antiaromatic are considered as non aromatic.
Stability of the compounds follows the following order:
Aromatic > Non-aromatic> Anti-aromatic
Note:
Carefully count the number of $\pi $ electrons in each compound. One lone pair of electrons is counted as 2$\pi $ electrons and one double bond or triple bond in a ring also contributes 2$\pi $ electrons.
Complete answer:
Aromatic compounds must obey Huckel’s rule and contain a certain number of $\pi $ electrons which should be equal to 4n+2. Here, n can be zero or any other positive integer. Therefore, compounds containing$2\pi $, $6\pi $, $10\pi $, $14\pi $ and so on electrons are aromatic.
Let us try to find the ring compound which obeys Huckel’s rule form the above given options.
${{C}_{4}}H_{4}^{-1}$
Possible ring structures with ${{C}_{4}}H_{4}^{-1}$ are shown below:
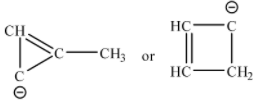
Neither of the compounds follow Huckel’s rule. In the three-membered ring compound the total number of $\pi $ electrons is 4, which is not a multiple of (4n+2)$\pi $. Ring compounds containing $4\pi $ electrons are anti-aromatic in nature. In the four-membered ring, there is a $s{{p}^{3}}$ hybridized tetrahedral carbon, so the ring is no longer planar. Thus, Huckel’s rule cannot be applied.
${{C}_{4}}H_{4}^{+1}$
Possible structure for ${{C}_{4}}H_{4}^{+1}$ is:

This ring is not planar as it contains a $s{{p}^{3}}$ hybridized carbon. Therefore, Huckel’s rule is not applicable.
${{C}_{4}}H_{4}^{-2}$
With the given molecular formula the possible ring structure is shown below:

This ring compound contains $6\pi $ electrons which is equal to (4n+2)$\pi $ when n=1. Therefore, it obeys Huckel’s rule and is aromatic.
${{C}_{4}}{{H}_{4}}$
Structure of ${{C}_{4}}{{H}_{4}}$ is given as:

This cyclic compound contains $4\pi $ electrons and thus, it is antiaromatic. This compound also does not follow Huckel’s rule.
The only compound following Huckel’s rule is ${{C}_{4}}H_{4}^{-2}$.
Hence, the correct option is (C).
Additional information:
Compounds which lacks one or more of the four requirements, i.e. cyclic, planar, conjugated and (4n+2)$\pi $ electrons for aromatic; 4n$\pi $ for antiaromatic, to be aromatic or antiaromatic are considered as non aromatic.
Stability of the compounds follows the following order:
Aromatic > Non-aromatic> Anti-aromatic
Note:
Carefully count the number of $\pi $ electrons in each compound. One lone pair of electrons is counted as 2$\pi $ electrons and one double bond or triple bond in a ring also contributes 2$\pi $ electrons.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




