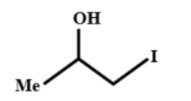
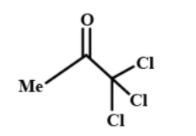
Which of the following gives yellow precipitate with NaOI?
(A)
(B)


Answer
602.7k+ views
Hint: This test is called an iodoform test. The final product of the substance has a medicinal smell like that of antiseptic solutions
Complete step by step solution:
As per the question asked, the yellow precipitate formed is due to the formation of iodoform or called triiodomethane. This reaction is a test for molecules having the groups like$R-CO-C{{H}_{3}}$$R-CO-C{{H}_{3}}$,$R-CH(OH)-C{{H}_{3}}$
-When ${{I}_{2}}$ reacts with NaOH it produces NaOI, and this on reaction with the compounds containing the groups like $R-CO-C{{H}_{3}}$, $R-CH(OH)-C{{H}_{3}}$ will give yellow precipitate as a result of iodoform formation.
And this is used as a test for ketones and aldehydes, secondary alcohol groups which can be converted to carbonyl group by oxidation also shows positive results towards this test.
The secondary alcohol must have at least a methyl group in the alpha position to show this test.
Description of Iodoform test- To carry out this test, first iodine and NaOH is warmed, which produces NaOI and this is allowed to pass through aldehydes and ketones containing $R-CO-C{{H}_{3}}$ and through secondary alcohol which has at least one methyl group in the alpha position . And we will observe a yellow precipitate with an antiseptic smell.
$R-CO-C{{H}_{3}}\xrightarrow[NaOH]{{{I}_{2}}}RCO{{O}^{-}}N{{a}^{+}}+CH{{I}_{3}}$
$R-CH\left( OH \right)-C{{H}_{3}}\xrightarrow[NaOH]{{{I}_{2}}}RCO{{O}^{-}}N{{a}^{+}}+CH{{I}_{3}}$
If we are testing for an aldehyde and the test is positive then it will be acetaldehyde as it is the only group having $C{{H}_{3}}CO$ group.
In the options above the first, option (A) is a secondary alcohol which has $R-CH(OH)-C{{H}_{3}}$ group and gets oxidized to carbonyl group during the reaction to give the final yellow precipitate.
So the correct answer for this question is option (a).
Note: In the mechanism of the iodoform reaction, first an acidic hydrogen is removed from the aldehyde, ketone or secondary alcohol. So there must be an acidic hydrogen available other than the hydrogen available in the methyl group which is attached to the carbonyl group that is why the second option given in the question gives a negative test result. Compounds which show positive results towards the iodoform test are – dimethyl ketone, acetaldehyde, secondary alcohols with methyl group in alpha position, ethanol etc.
Complete step by step solution:
As per the question asked, the yellow precipitate formed is due to the formation of iodoform or called triiodomethane. This reaction is a test for molecules having the groups like$R-CO-C{{H}_{3}}$$R-CO-C{{H}_{3}}$,$R-CH(OH)-C{{H}_{3}}$
-When ${{I}_{2}}$ reacts with NaOH it produces NaOI, and this on reaction with the compounds containing the groups like $R-CO-C{{H}_{3}}$, $R-CH(OH)-C{{H}_{3}}$ will give yellow precipitate as a result of iodoform formation.
And this is used as a test for ketones and aldehydes, secondary alcohol groups which can be converted to carbonyl group by oxidation also shows positive results towards this test.
The secondary alcohol must have at least a methyl group in the alpha position to show this test.
Description of Iodoform test- To carry out this test, first iodine and NaOH is warmed, which produces NaOI and this is allowed to pass through aldehydes and ketones containing $R-CO-C{{H}_{3}}$ and through secondary alcohol which has at least one methyl group in the alpha position . And we will observe a yellow precipitate with an antiseptic smell.
$R-CO-C{{H}_{3}}\xrightarrow[NaOH]{{{I}_{2}}}RCO{{O}^{-}}N{{a}^{+}}+CH{{I}_{3}}$
$R-CH\left( OH \right)-C{{H}_{3}}\xrightarrow[NaOH]{{{I}_{2}}}RCO{{O}^{-}}N{{a}^{+}}+CH{{I}_{3}}$
If we are testing for an aldehyde and the test is positive then it will be acetaldehyde as it is the only group having $C{{H}_{3}}CO$ group.
In the options above the first, option (A) is a secondary alcohol which has $R-CH(OH)-C{{H}_{3}}$ group and gets oxidized to carbonyl group during the reaction to give the final yellow precipitate.
So the correct answer for this question is option (a).
Note: In the mechanism of the iodoform reaction, first an acidic hydrogen is removed from the aldehyde, ketone or secondary alcohol. So there must be an acidic hydrogen available other than the hydrogen available in the methyl group which is attached to the carbonyl group that is why the second option given in the question gives a negative test result. Compounds which show positive results towards the iodoform test are – dimethyl ketone, acetaldehyde, secondary alcohols with methyl group in alpha position, ethanol etc.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




