The structure of toluene is?
A.
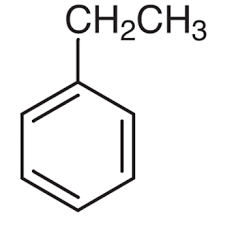
B.
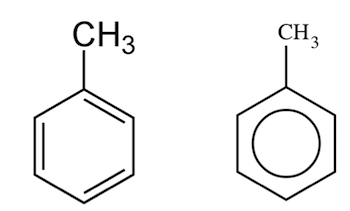
C.
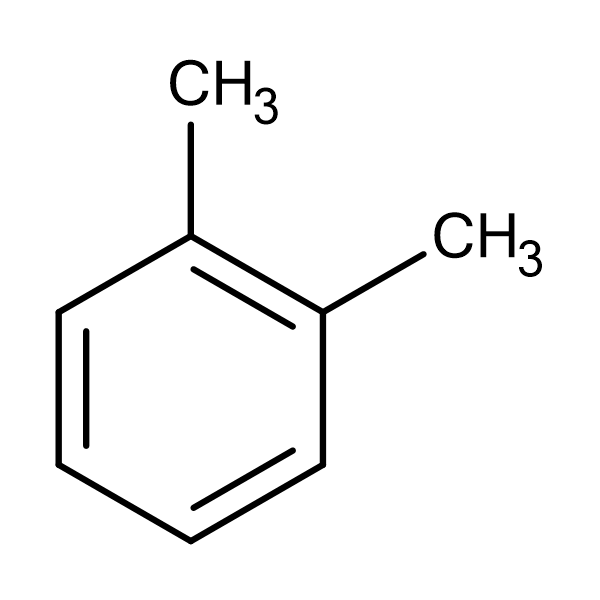
D.
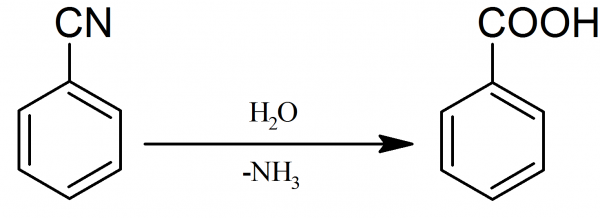




Answer
638.7k+ views
Hint: Toluene is a mono-substituted benzene derivative and it is more reactive than benzene toward electrophiles. We can use properties to identify the correct structure.
Complete step by step answer:
Toluene is an aromatic hydrocarbon. It is a colorless, water-insoluble liquid with the smell associated with paint thinners.
It is a mono-substituted benzene derivative, consisting of a methyl group attached to a phenyl group. As such, its IUPAC systematic name is methylbenzene.
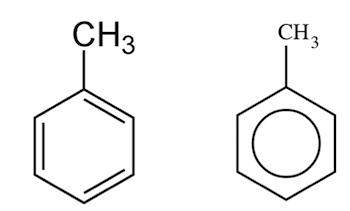
Which is given in option B .
We can also identify structures provided in other options too.
The structure in option A, is of ethylbenzene. It is also a mono-substituted benzene derivative.
The structure in option C, is of 1,2-dimethylbenzene. It is a di-substituted benzene derivative.
The structure in option D, is of benzoic acid. It is also a mono-substituted benzene derivative.
Thus, the correct option is B.
Additional information: Toluene is used as the solvent in some types of paint thinner, permanent markers, contact cement and certain types of glue.
Toluene is sometimes used as a recreational inhalant and has the potential of causing severe neurological harm.
Note: Toluene reacts as a normal aromatic hydrocarbon in electrophilic aromatic substitution. Because the methyl group has greater electron-releasing properties than a hydrogen atom in the same position, toluene is more reactive than benzene toward electrophiles.
Complete step by step answer:
Toluene is an aromatic hydrocarbon. It is a colorless, water-insoluble liquid with the smell associated with paint thinners.
It is a mono-substituted benzene derivative, consisting of a methyl group attached to a phenyl group. As such, its IUPAC systematic name is methylbenzene.

Which is given in option B .
We can also identify structures provided in other options too.
The structure in option A, is of ethylbenzene. It is also a mono-substituted benzene derivative.
The structure in option C, is of 1,2-dimethylbenzene. It is a di-substituted benzene derivative.
The structure in option D, is of benzoic acid. It is also a mono-substituted benzene derivative.
Thus, the correct option is B.
Additional information: Toluene is used as the solvent in some types of paint thinner, permanent markers, contact cement and certain types of glue.
Toluene is sometimes used as a recreational inhalant and has the potential of causing severe neurological harm.
Note: Toluene reacts as a normal aromatic hydrocarbon in electrophilic aromatic substitution. Because the methyl group has greater electron-releasing properties than a hydrogen atom in the same position, toluene is more reactive than benzene toward electrophiles.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




