The oxidation states of S atoms in ${S_4}{O_6}^{2 - }$ from left to right respectively, are:
A. +6, 0, 0, +6
B. +3, 1, +1, +3
C. +5, 0, 0, +5
D. None of the above
Answer
547.4k+ views
Hint: To solve this question, first we need to understand the meaning of oxidation state. An oxidation is the process which determines what part of the reaction is being oxidized and what part is being reduced in a redox reaction.
Complete step by step answer:
As we know that an oxidation state refers to two things:
Oxidation as well as reduction in terms of electron transfer occurs in a redox reaction and electron-half-equations
Now, let’s consider the structure of ${S_4}{O_6}^{2 - }$
Then, we see that in the middle two sulfur have 0 oxidation states as an atom which is bonded with similar atoms has an oxidation state of 0. So, the total oxidation state of sulfur in the compound is 10. Then, the oxidation state of the leftmost and the rightmost sulfur is +5, as oxygen is more electronegative.
Therefore, the oxidation state of sulfur is n − 2 − 2 − 1+ 0 = 0; n = 5
So, the oxidation state of sulfur is +5 isolated S−S linkage have zero oxidation state.
⇒Thus, the oxidation state becomes +5, 0, 0, + 5.
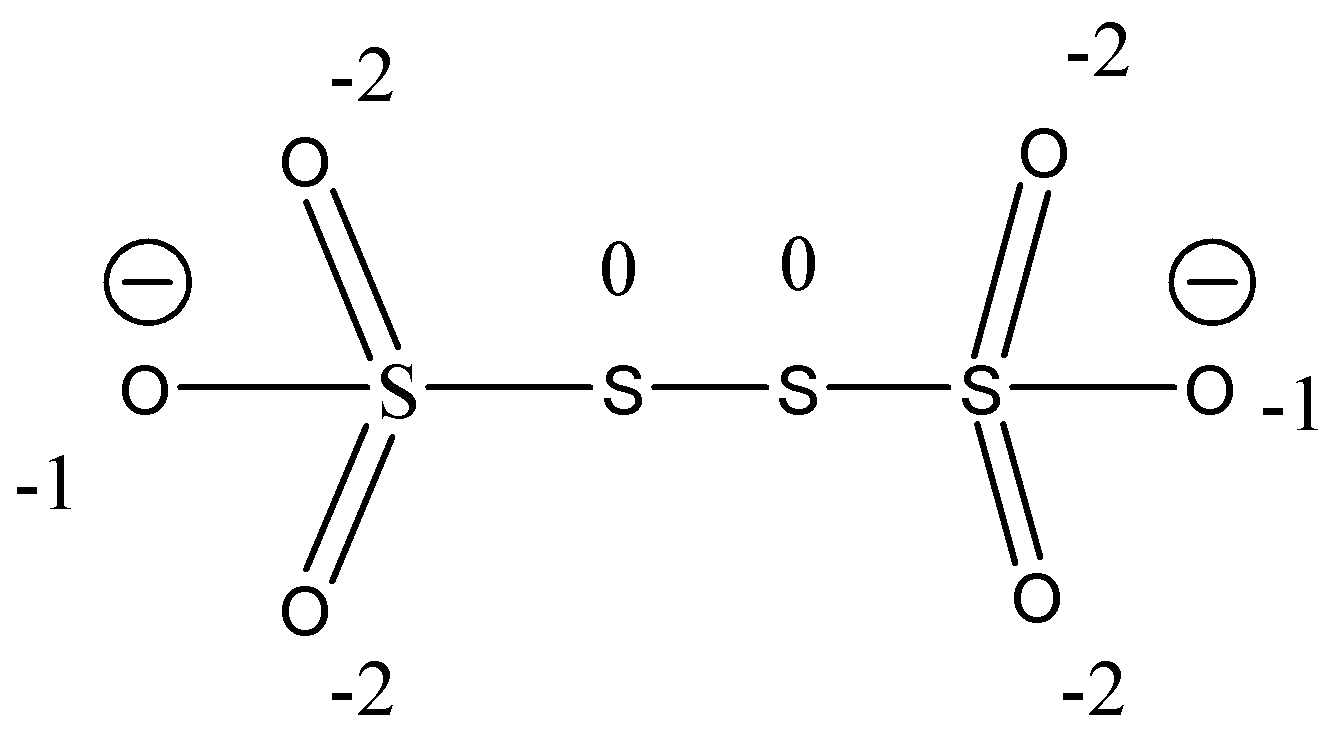
$\therefore $The option C is correct answer.
Note:
We need to remember that in oxoacids, sulfur shows a tetrahedral structure with respect to oxygen. And oxoacids are the acids that contain oxygen. The oxoacids have a minimum of one \[S = O\]bond and one \[S - OH\]bond. Also, there are terminal peroxide groups, terminal\[S = S\], terminal and bridging oxygen atoms in these oxoacids.
Complete step by step answer:
As we know that an oxidation state refers to two things:
Oxidation as well as reduction in terms of electron transfer occurs in a redox reaction and electron-half-equations
Now, let’s consider the structure of ${S_4}{O_6}^{2 - }$
Then, we see that in the middle two sulfur have 0 oxidation states as an atom which is bonded with similar atoms has an oxidation state of 0. So, the total oxidation state of sulfur in the compound is 10. Then, the oxidation state of the leftmost and the rightmost sulfur is +5, as oxygen is more electronegative.
Therefore, the oxidation state of sulfur is n − 2 − 2 − 1+ 0 = 0; n = 5
So, the oxidation state of sulfur is +5 isolated S−S linkage have zero oxidation state.
⇒Thus, the oxidation state becomes +5, 0, 0, + 5.

$\therefore $The option C is correct answer.
Note:
We need to remember that in oxoacids, sulfur shows a tetrahedral structure with respect to oxygen. And oxoacids are the acids that contain oxygen. The oxoacids have a minimum of one \[S = O\]bond and one \[S - OH\]bond. Also, there are terminal peroxide groups, terminal\[S = S\], terminal and bridging oxygen atoms in these oxoacids.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




