The number of chiral centers in open-chain structures of glucose is
A.3
B.4
C.5
D.6
Answer
609k+ views
Hint: We know that the chiral center is a point at which all the four groups surrounding the point are of different atoms. An example of it is $C(F)(Cl)(Br)H$. The molecular formula of Glucose is
${{{C}}_{{6}}}{{{H}}_{{{12}}}}{{{O}}_{{6}}}$. From the open chain structure of glucose, the number of chiral centers present in the glucose molecule can be found out.
Complete step by step answer:
As we know that here, the chiral center is a carbon that is attached to four different groups of atoms.
The open-chain structure of glucose is drawn as -
.
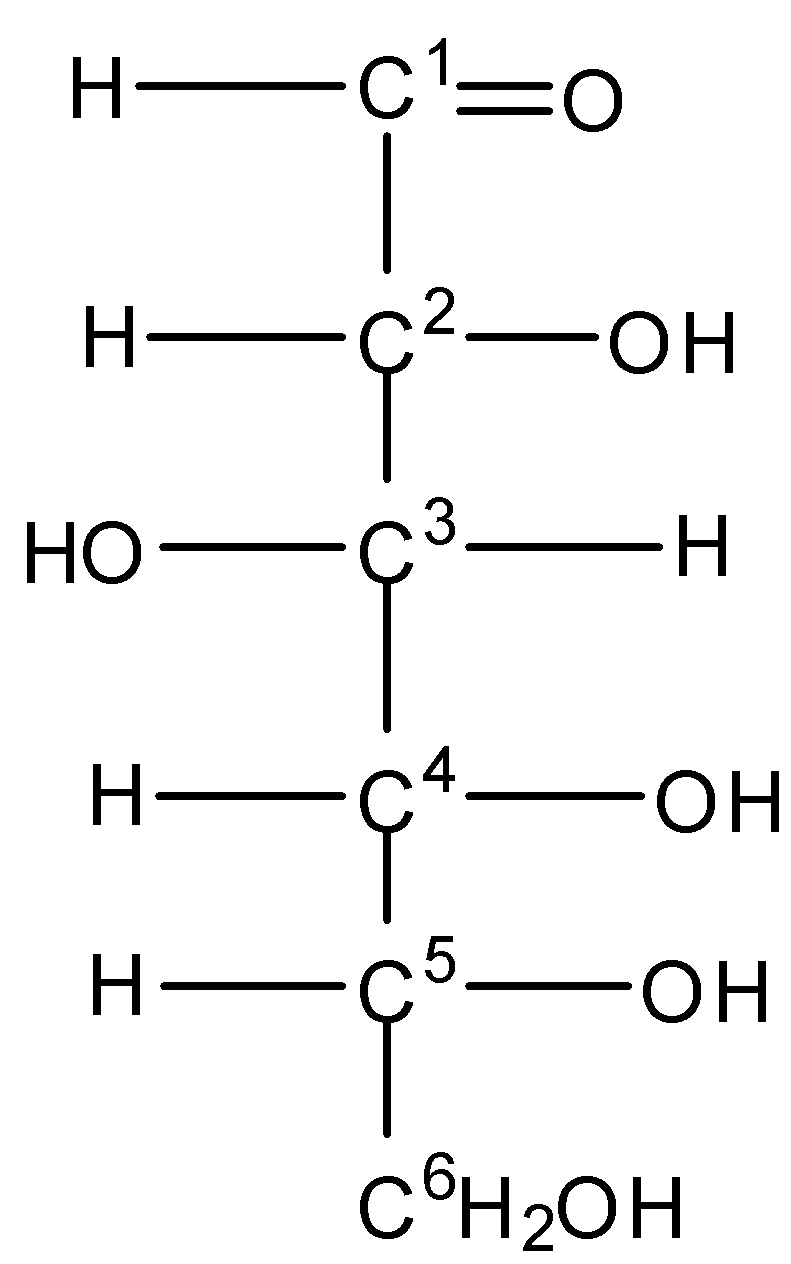
Here we can see that there is no internal plane of symmetry.
In ${C^1}$, there is a double bond to Oxygen, so we can consider it a 2 oxygen atom. So it is not a chiral carbon.
On checking the ${C^2},{C^3},{C^4},{C^5}$ , the valency of carbon is satisfied by 4 different groups of atoms. Hence they are chiral.
Therefore the total number of chiral centers present in the open-chain structure of glucose is four.
Therefore, the atomic masses of the element X and Y are 25.6 and 42.6
So, the correct answer is Option B.
Note: The open-chain structure of glucose has 4 chiral centers. The cyclic structure of glucose resembles pyran and due to the formation of cyclic hemiacetal, there are five chiral centers present in the cyclic form of glucose.
${{{C}}_{{6}}}{{{H}}_{{{12}}}}{{{O}}_{{6}}}$. From the open chain structure of glucose, the number of chiral centers present in the glucose molecule can be found out.
Complete step by step answer:
As we know that here, the chiral center is a carbon that is attached to four different groups of atoms.
The open-chain structure of glucose is drawn as -
.

Here we can see that there is no internal plane of symmetry.
In ${C^1}$, there is a double bond to Oxygen, so we can consider it a 2 oxygen atom. So it is not a chiral carbon.
On checking the ${C^2},{C^3},{C^4},{C^5}$ , the valency of carbon is satisfied by 4 different groups of atoms. Hence they are chiral.
Therefore the total number of chiral centers present in the open-chain structure of glucose is four.
Therefore, the atomic masses of the element X and Y are 25.6 and 42.6
So, the correct answer is Option B.
Note: The open-chain structure of glucose has 4 chiral centers. The cyclic structure of glucose resembles pyran and due to the formation of cyclic hemiacetal, there are five chiral centers present in the cyclic form of glucose.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




